Electromagnetic Spectrum: The sun is our planet’s principal source of energy, and its energy travels in the form of electromagnetic radiation. Electromagnetic energy moves across space at the speed of light in the form of waves of electric and magnetic fields with a range of frequencies or wavelengths.
Electromagnetic radiation is a common occurrence in our daily lives. All electromagnetic waves, from visible light, that our eyes can detect to microwave radiation that heats our meals or radio waves that power our radios, X-rays that enable doctors to identify any injury in our bones, or UV radiation emitted by a hot surface, are EM waves.
In this article, we have provided all types of electromagnetic radiation in the Electromagnetic Spectrum, their properties, diagrams, and applications of Electromagnetic Spectrum.
What is an Electromagnetic Spectrum?
Consider the electromagnetic spectrum as a wide cosmic highway that facilitates the movement of various kinds of light. There is a vast spectrum of energy waves present in this, ranging from extremely short waves like X-rays to long, languid waves like radio broadcasts. This spectrum extends beyond the light that our eyes can detect.
The tiny portion of light that we can see, known as visible light, which includes every color in the rainbow, is located exactly in the middle of this enormous spectrum. But there’s so much more to it than that. On one extreme are the incredibly small and intense gamma and X-rays, which are employed in medicine to view the body and can penetrate through solid objects. The radio waves, on the other hand, are like the long, slow waves that carry your favorite radio station’s music through the air to your radio.
Electromagnetic Spectrum Definition
The electromagnetic (EM) spectrum is the range of all types of electromagnetic radiation, which is energy that disperses and expands as it travels. This includes everything from the visible light emitted by a household lamp to the radio waves broadcast by radio stations, illustrating the diverse forms of electromagnetic radiation.
Electromagnetic Waves
Waves created by the interaction of vibrating electric and magnetic fields are known as electromagnetic waves. An oscillating electric and magnetic field makes up EM waves.
A charged particle, in general, produces an electric field. This electric field exerts a push on other charged particles. Positive charges accelerate in the field’s direction, whereas negative charges accelerate in the opposite direction of the field. A traveling charged particle creates a magnetic field. This magnetic field exerts a push on other moving particles. Because the force acting on these charges is always perpendicular to their movement, it only affects the direction of the velocity, not the speed. As a result, a speeding charged particle generates an electromagnetic field. Electromagnetic waves are nothing more than electric and magnetic fields traveling at the speed of light c through open space.
When a charged particle oscillates about an equilibrium location, it is said to be accelerating. If the charged particle’s oscillation frequency is f, it creates an electromagnetic wave of frequency f. The wavelength of this wave may be calculated using the formula:
λ = c/f
Electromagnetic waves are a type of energy transfer that occurs in space.
Representation of Electromagnetic Waves
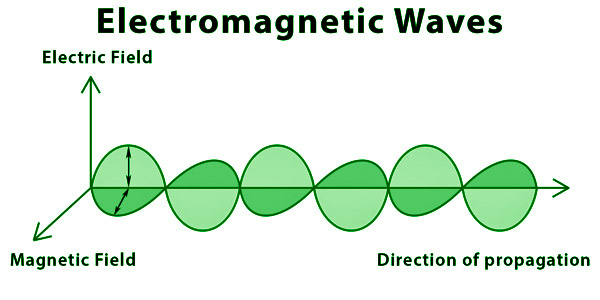
Electromagnetic Spectrum
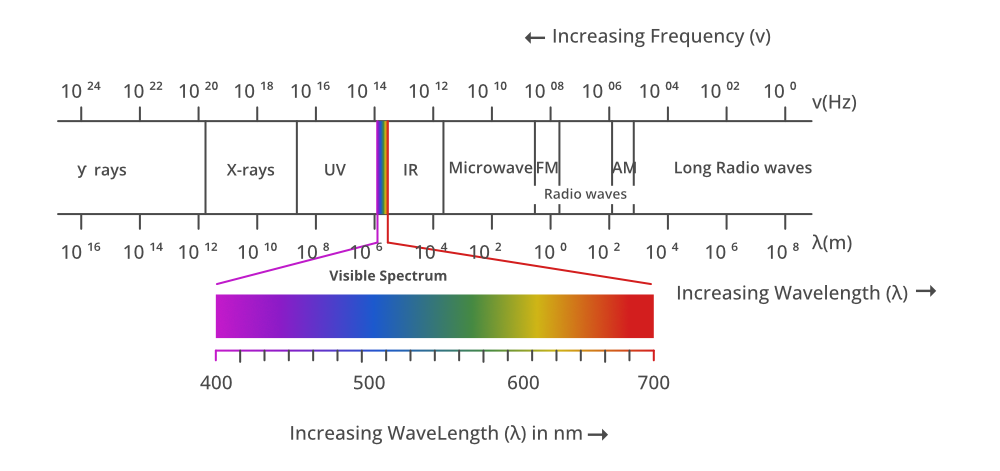
The electromagnetic spectrum is a collection of frequencies, wavelengths, and photon energies of electromagnetic waves spanning from 1Hz to 1025Hz, equivalent to wavelengths ranging from a few hundred kilometres to a size smaller than the size of an atomic nucleus. The electromagnetic spectrum can thus be described as the range of all types of electromagnetic radiation in basic terms. In a vacuum, all electromagnetic waves travel at the same speed as light. For different forms of electromagnetic waves, however, the wavelengths, frequencies, and photon energy will vary.
Electromagnetic Spectrum Diagram
The frequency (f), wavelength (λ), energy (E) of an electromagnetic wave are related to each other as:
λ=c/f
f=E/h
E=hc/λ
where
- c=3×108m/s represents the speed of light in a vacuum
- h=6.626×10–34J.s represents Planck’s constant.
Electromagnetic Waves in Electromagnetic Spectrum
Radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays, gamma rays, and cosmic rays make up the full range (electromagnetic spectrum) in decreasing order of frequency and rising order of wavelength.
- The rapid travel of charged particles across conducting wires causes these waves.
- Radio, television, and telecom signals are transmitted through them.
- These waves have a frequency range of around 3kHz to 300MHz.
- In the ultrahigh-frequency (UHF) band, cellular phones employ radio waves to convey voice communication.
- Radio picks up radio waves that are broadcast by radio stations. Radio waves can be emitted by gases and stars in space. The majority of radio waves are used for TV and mobile communication.
Microwaves
- Microwaves are a type of electromagnetic radiation that has a frequency of a few gigahertz (GHz).
- Klystrons, magnetrons, and Gunn diodes are unique vacuum tubes that produce them.
- Microwaves are commonly utilised in aviation navigation due to their short wavelengths.
- These rays are employed in microwaves, which aid in the heating of meals in homes and offices. It’s also used by astronomers to figure out and understand the structure of surrounding galaxies and stars.
Infrared Rays
- Infrared waves are produced by hot bodies and molecules and are thus referred to as heatwaves.
- Infrared rays are near the low-frequency or long-wavelength end of the visible light spectrum.
- The greenhouse effect caused by these rays is critical for maintaining global warming and average temperatures.
- Greenhouse gases such as carbon dioxide and water vapour trap these radiations in the earth’s atmosphere.
- Night vision goggles make use of these radiations. Infrared light generated by objects in the dark can be read and captured by these devices. Infrared light is used to trace interstellar dust in space. Infrared radiation is emitted by electronic devices and is commonly employed in remote switches for a variety of household gadgets.
Visible Rays
- Visible rays are electromagnetic waves that can be seen with the naked eye. They are the most common type of electromagnetic waves.
- These can be found in the frequency range of 4×1014Hz–7×1014Hz or the wavelength range of 400nm–700nm.
- The visible light rays reflected or released from the objects around us assist us in seeing the world, and the range of visible radiation is different for different creatures.
- Devices that emit light in the visible area of the electromagnetic spectrum include bulbs, lamps, candles, LEDs, tube lights, and so on.
Ultraviolet Rays
- Although the sun is the primary source of ultraviolet radiation on Earth, the ozone layer absorbs the majority of UV energy before it reaches the atmosphere.
- UV radiation has a wavelength of 400nm–1nm.
- These radiations are emitted by special lamps and extremely hot bodies, and in big numbers, they can cause significant injury to humans. It tans the skin and creates burns.
- Because these radiations may be focused on tiny beams, they are used in high precision applications such as LASIK or laser-based eye surgery.
- UV lamps are used in water purifiers to eliminate microorganisms that may be present in the water.
- When working with UV welding arcs, welders use special goggles to protect their eyes.
- This electromagnetic radiation is found outside of the ultraviolet (UV) region of the electromagnetic spectrum and is extremely valuable in the medical field.
- The wavelength range of X-ray radiation is 1nm–10–3nm.
- By blasting a metal target with high-energy electrons, X-rays can be produced.
- X-rays are a diagnostic technique in medicine that can be quite helpful in the treatment of some types of cancer. To find the source of the problem, a doctor utilises an x-ray scanner to scan our bones or teeth. Overexposure to x-rays can cause harm or death to the organism’s healthy tissues. As a result, extreme caution must be exercised when dealing with x-rays.
- At the airport checkpoint, security agents utilise it to search through passengers’ luggage. X-rays are also emitted by the universe’s heated gases.
Gamma-Rays
- The universe is the largest gamma-ray generator.
- These rays are in the electromagnetic spectrum‘s higher frequency region.
- Gamma rays have wavelengths ranging from 10–12m to 10–14m.
- Radioactive nuclei release high-frequency radiations, which are also created during nuclear processes.
- Gamma rays have a wide range of medical applications, including the destruction of cancerous cells. Gamma-ray imaging is a technique used by doctors to examine the insides of patients’ bodies.
Electromagnetic Spectrum Wavelength and Frequency Table
The table provided below provides the Electromagnetic Spectrum Wavelength and Frequency of all types of electromagnetic radiations:
|
Types of Radiation
|
Frequency range (Hz)
|
Wavelength Range
|
|
Gamma-rays
|
1020-1024
|
<10-12 m
|
|
X-rays
|
1017-1020
|
1 nm – 1 pm
|
|
Ultraviolet rays
|
1015-1017
|
400 nm – 1 nm
|
|
Visible rays
|
4 x 1014 – 7.5 x 1014
|
750 nm – 400 nm
|
|
Near-infrared
|
1 x 1014 – 4 x 1014
|
2.5 μm – 750 nm
|
|
Infrared rays
|
1013 – 1014
|
25 μm – 2.5 μm
|
|
Microwaves
|
3 x 1011 – 1013
|
11 mm – 25 μm
|
|
Radio waves
|
< 3x 1011
|
>1 mm
|
Also Read:
Spectroscopy
In terms of wavelength or frequency, spectroscopy is a method for determining the emission and absorption of light and other radiation as it interacts with matter.
As a ray of light passes through matter, it is scattered. It interacts with atoms and molecules of the given substance, and these atoms interact with light waves of similar frequencies based on their resonance frequencies. When light rays collide with an atom in an excited state, certain distinctive frequencies are released, resulting in a line spectrum. This line spectrum is made up of a collection of emission lines that isn’t continuous. The wavelengths of the light produced are separated. When light with continuous wavelengths is passed through a low-density material, an absorption spectrum is created. Atoms and molecules with characteristic frequencies similar to light waves will be absorbed, resulting in a continuous spectrum with a few lines missing.
Applications / Uses of Electromagnetic Spectrum
The presence of the full electromagnetic spectrum was originally demonstrated by Maxwell. His mathematics suggested that electromagnetic radiation may have an endless number of frequencies. The electromagnetic spectrum is a frequency and wavelength-based organisation of various radiations. The following are some examples of EM-spectrum applications:
- Hertz was the first to discover radio waves and microwaves. Wireless television and radio, as well as mobile communication, arose as a result of these waves.
- Ultraviolet radiation is useful for the ionisation of atoms, which aids in the initiation of numerous chemical reactions.
- The gamma rays were discovered by Paul Villard. These are employed in the development of nuclear medicine and ionisation experiments.
- X-rays were invented by Roentgen. These are used to discover problems with the bones and teeth, as well as abnormalities.
- The visible light portion of the electromagnetic spectrum allows us to see the world around us. This part of the electromagnetic spectrum aids in the perception of all objects, including colours.
People Also View:
Electromagnetic Spectrum – Sample Questions
Question 1: What are electromagnetic waves?
Answer:
Electromagnetic waves are vibrations made up of perpendicularly oscillating electric and magnetic fields.
Question 2: What are the frequency and wavelength of an EM wave of energy 6.626 x 10-19 J?
Answer:
Frequency(f) = E/h
= 1015 Hz.
Wavelength(λ) = c/f
= 3 x 108 / 1015
= 3 x 107 m
Question 3: What is Spectroscope?
Answer:
Spectroscopy is the study of how light and other electromagnetic radiation are emitted and absorbed by matter based on the wavelength or frequency of the energy.
Summary – Electromagnetic Spectrum
Consider the electromagnetic spectrum as an enormous rainbow of energy that is invisible to the human eye, where each color stands for a distinct kind of wave that travels through space. This spectrum is the universe’s means of transmitting energy throughout space, from the long, smooth waves of radio transmissions to the tiny, potent bursts of gamma rays. These waves are created when magnetic and electric forces combine to travel through space at the speed of light. They can be found in everything around us, including the sun’s warmth on our skin, X-rays that can see inside our bodies, microwaves that cook our food, UV rays that tan us, and radio waves that play music for us to hear. Every kind of wave in this cosmic rainbow has its unique wavelength and frequency, forming a bridge between the energy of the stars and the everyday technologies that power our world.
FAQs on Electromagnetic Spectrum
What is the electromagnetic spectrum?
The electromagnetic spectrum is a range of all types of electromagnetic radiation, including visible light, radio waves, and X-rays, organized by their wavelengths.
What are the 7 electromagnetic waves?
The seven electromagnetic waves are radio waves, microwaves, infrared, visible light, ultraviolet, X-rays, and gamma rays.
What is electromagnetic spectrum and its functions?
The electromagnetic spectrum is a range of all types of electromagnetic radiation, organized by wavelength. Its functions include transmitting information (like radio waves), heating (like microwaves), and imaging (like X-rays).
What are the Practical Applications of Electromagnetic Waves?
Practical applications of electromagnetic waves include communication (e.g., radio and Wi-Fi), cooking (e.g., microwaves), medical imaging (e.g., X-rays), and more.
Which electromagnetic spectrum has the highest wavelength?
Radio waves in the electromagnetic spectrum have the highest wavelength.
Which part of the electromagnetic spectrum has the highest frequency?
Gamma rays in the electromagnetic spectrum have the highest frequency.
Which part of the electromagnetic spectrum carries the least energy?
Radio waves in the electromagnetic spectrum carry the least energy.
Where in the electromagnetic spectrum is visible light?
Visible light is in the middle of the electromagnetic spectrum.
What is the increasing frequency of electromagnetic spectrum?
The electromagnetic spectrum increases in frequency from radio waves to microwaves, then infrared, visible light, ultraviolet, X-rays, and finally, gamma rays, with gamma rays having the highest frequency.
What is emw spectrum?
The EMW (Electromagnetic Wave) spectrum, also known as the electromagnetic spectrum, is the complete range of all electromagnetic waves, ordered by their wavelengths and frequencies. It includes a wide range of waves, from low-frequency radio waves to high-frequency gamma rays, encompassing everything from radio and microwaves to visible light, X-rays, and beyond. Each type of wave has unique properties and applications in science, technology, and everyday life.
What is electromagnetic waves frequency?
Electromagnetic waves vary in frequency, which refers to how many wave cycles occur in a given time period. Frequency is typically measured in hertz (Hz), where one hertz represents one wave cycle per second. Electromagnetic waves cover a vast range of frequencies, with radio waves having lower frequencies (typically measured in megahertz or gigahertz) and gamma rays having extremely high frequencies (typically measured in terahertz or petahertz). The frequency of electromagnetic waves is inversely related to their wavelength, meaning higher frequency waves have shorter wavelengths, and vice versa.
Share your thoughts in the comments
Please Login to comment...