Condensation
Last Updated :
25 Apr, 2024
Condensation is the process in which water vapour cools down to become liquid. Condensation in Chemistry can be viewed as the reverse process of evaporation, where liquid water heats and acquires vapour form. Condensation occurs when any of the two conditions come true, i.e., the air is cooled to its dew point or it becomes so saturated with water vapour that it cannot hold any more water. Condensation transforms water vapour into liquid water.
In this article, we will learn what is condensation, examples of condensation, dew point, water cycle, applications of condensation and condensation vs evaporation in detail.
What is Condensation?
Condensation is the process of change of the phase of water where the vapour transforms to a liquid state. Condensation can be achieved in one of the following two ways :
- Cooling of air until its dew point.
- Saturation with water vapour until the point that it cannot hold any further quantity of water.
Condensation is considered to be the reverse reaction of evaporation, where the liquid water turns to a vapour state. The rate of condensation can be measured through the process of evaporation into the air moisture at various atmospheric pressures and temperatures. The process of condensation involves the reaction where two molecules are combined together accompanied by the loss of water droplets. It also involves the removal of heat from a system where the vapour is converted into liquid.
Condensation Definition
Alternatively, Condensation can also be defined as,
- Liquification of gas is also considered Condensation.
- Reaction which results in the loss of water molecules is considered to be Condensation.
- If the heat from a system is removed in such a manner that vapour is converted into a liquid it also comes under Condensation.
Check: Real Life Applications of Condensation
Condensation Process
Condensation is a physical process in which water is changed from its vapour form to its liquid form. Water in its vapour form is hot (about 100℃) and cooling the water vapours below its boiling point is called condensation process. In condensation process water vapour cools down immediately and water droplets are formed.
We all have seen water droplets that are formed outside a cold-water bottle in a summer afternoon these are formed by the process of condensation. Apart form this the water droplet on the lid of boling cattel is formed by condensation process.
Examples of Condensation
Condensation is pretty common and we see condensation in our day-to-day life.
- Water droplets on a bottle of cold water on a humid day.
- Droplets of water on the spectacles in winter, etc.
These are examples of condensation which are experienced by us daily.
Condensation also forms fog in winter, the water covering the inside of a window on a cold day, and the moisture on the inside of car windows all are examples of condensation. In all the examples discussed above water leaves the vapour state in the warm air and condenses into liquid as it cools.
Condensation – Water Cycle
Condensation helps the formation of clouds in the atmosphere. Water vapour present in the atmosphere is responsible for the formation of clouds which results in rain. Molecules of water are in constant movement between the different states of matter– i.e. solid, liquid, or gas. The vapours present in the air are loosely bound to each other and are in a state of constant random motion.
When excess moisture collects in the air water molecules become more organized. As an effect of this heat is released into the atmosphere accompanied by the phase change from vapour to liquid. The warm air first rises up and then cools down in the form of rain. The image added below shows the water cycle,
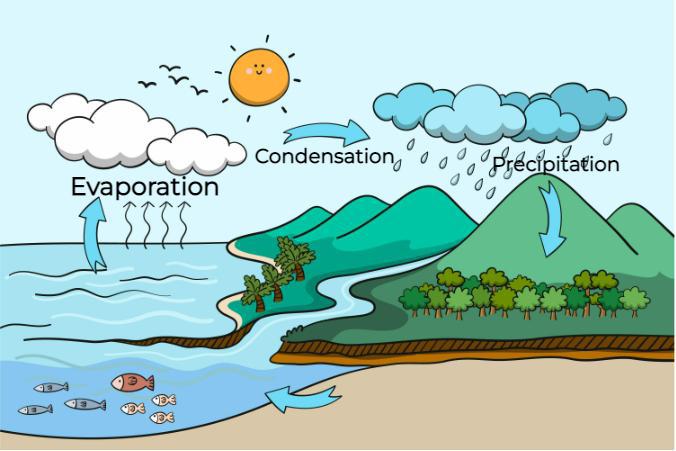
Condensation in the atmosphere occurs only when the atmosphere is fully saturated with water vapours. Dust particles, smoke particles and bacteria present in the atmosphere allow water vapour to condense. This water vapour condenses and carries water to different surfaces and showers them in the form of rain.
Check: Aldol Condensation
Dew Point
Dew is the water condensed in the atmosphere. The temperature at which the process of condensation occurs is known as the dew point. There are various applications visible with respect to the dew point:
- Cars and houses are mostly covered with water droplets early in the morning. This is because the air temperature at night stoops down below the dew point naturally.
- There are water droplets observed outside the surface of cold glass. This is because when the warm air outside comes in contact with the cold surface, it reaches its dew point and condenses.
- Forming of clouds in the atmosphere. Clouds have flat bottoms due to which the condensation starts. The flat bottom is the point where water vapour condenses to form water droplets.
Check: Step Growth or Condensation Polymerization
Why Condensation Occur?
Condensation occurs at high pressure and low temperature. Whenever the pressure of the liquid state of the gas is lesser than the pressure of the condensing gas, the process of condensation occurs. During this, there is the release of heat energy, resulting in the transformation of the state of matter.
The process easily takes place around dust particles or smoke or microscopic bacteria. The atmosphere should be fully saturated in order to attain the level of maximum vapour pressure.
Saturation
Clouds can be considered as a mass of water droplets formed in the atmosphere. The water particles are situated far from each other in the atmosphere. Whenever more droplets combine in the clouds, they attain the point of saturation with the water vapour. Whenever more water droplets collect in the clouds, they become saturated. They cannot contain any more water vapour on increasing the density. This increases the closeness of the water molecules. This results in the condensation of water vapour and the formation of rain.
Warm climates are often considered to be more humid than cold weather. This is because cold air contains fewer vapours in comparison to warm air. The main reason behind this is that the water vapour remains in the air instead of condensing into the rain. In cold climates, condensation is easy.
Check: Types of Cloud
Applications of Condensation
- Formation of clouds and water cycle: Water balance in the environment is maintained by the process of condensation.
- Manufacturing of pure substances and separation of mixtures. Used in industrial processes.
- Morning Dew: Condensation of moisture present in the air and its condensation on grasses overnight.
- Fog on the mirror: After a hot shower, the shower moisture condenses on the cool mirror.
- Water droplets on the can.
- Clouds
- Foggy breath: Cold weathers allow your inner moisture to condense into large droplets.
- Foggy windshield: The moisture in the surrounding gets captured on the cold windshield, which condenses in the form of water droplets.
Difference between Evaporation and Condensation
The difference between condensation and evaporation is tabulated below:
|
Evaporation
|
Condensation
|
| Liquid water changes into vapour form in case of evaporation. |
Water vapour changes into liquid water in case of the the condensation. |
| In the process of Evaporation, energy is consumed. |
In the process of Condensation, energy is released. |
| In case of evaporation, a weak bond is formed between water molecules. |
In case of condensation, a strong bond is formed between water molecules. |
Read More,
Condensation – FAQs
What is Condensation Definition?
The process of conversion of gas form into its liquid form is called Condensation.
Example:
- Dew on the grass in the morning.
- Water droplets on the surface of cold water bottle.
What happens during Condensation?
Condensation is the process of converting water vapor into liquid water; it’s the opposite process of evaporation.
What are Examples of Condensation?
A few common example of condensation are:
- Water forming on the outer surface of a cold cup.
- Moisture on car windows during a cool night.
- Dew, fog, clouds, all are examples of condensation.
What are harmful effects of Condensation?
There are some serious side effects of condensation:
- Fungus and mildew growth
- Blistering or peeling of paint
- Damp spots on ceilings
What is the Cause of Condensation?
When the warm air comes in contact with the cooler surfaces the excess of the moisture undergoes condensation. The reason behind this is that the excess moisture can’t be held on the cooler surface.
Is Condensation Physcial Or Chemical Change?
As condensation is the process in which a gas changes to water, so in the condensation process only phycial changes occurs, thus, Condensation is a Physcial Change.
Share your thoughts in the comments
Please Login to comment...