Ozone layer depletion is the decline of the ozone layer in the upper atmosphere. This occurs when ozone molecules come into touch with and are destroyed by atoms of chlorine and bromine found in the atmosphere. Ozone molecules can be destroyed by one chlorine molecule. It degrades faster than it is produced. In this article, we will study the ozone layer, ozone depletion, causes and prevention of ozone layer depletion.
What is an Ozone Layer?
The area of the stratosphere that absorbs the majority of the Sun’s UV energy is known as the ozone layer or ozone shield. In comparison to other areas of the atmosphere, it has a high concentration of ozone (O3), yet it is still relatively low in comparison to other gases in the stratosphere. The average ozone concentration in the Earth’s atmosphere is about 0.3 parts per million, whereas the ozone layer has a concentration of fewer than 10 parts per million. Although its thickness varies seasonally and geographically, the ozone layer is primarily located in the lower stratosphere, from about 15 to 35 kilometers (9 to 22 mi) above Earth.
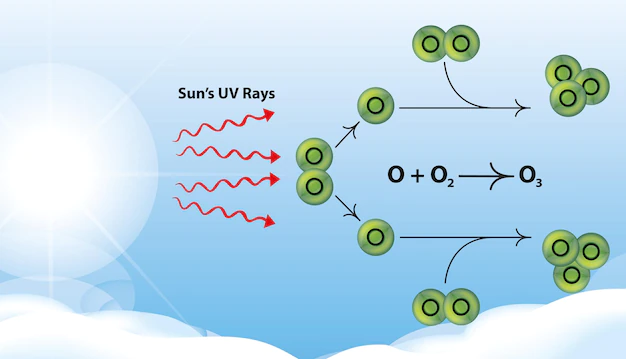
What is Ozone Layer Depletion?
The steady thinning of the ozone layer in the upper atmosphere of the earth is known as ozone layer depletion. The ozone hole, a far more important springtime drop in stratospheric ozone in Earth’s Polar Regions, is also brought on by ozone depletion. A decrease in the ozone layer in the high atmosphere is referred to as ozone layer depletion. It harms the environment and the natural world. For the atmosphere and all of the organisms on the planet, including the flora and wildlife, ozone layer depletion is a major problem.
Concern over increasing cancer risks and other harmful effects due to ozone depletion and the ozone hole has spread throughout the world. The ozone layer blocks harmful ultraviolet (UVB) light wavelengths from entering the Earth’s atmosphere. These wavelengths harm plants and animals as well as cause skin cancer, sunburn, permanent blindness, and cataracts, all of which were predicted to sharply increase as a result of the weakening of the ozone layer. The Montreal Protocol, which outlaws the manufacture of CFCs, halons, and other ozone-depleting substances, was adopted in 1987 as a result of these concerns. Scientists are currently working to create new refrigerants to replace current ones.
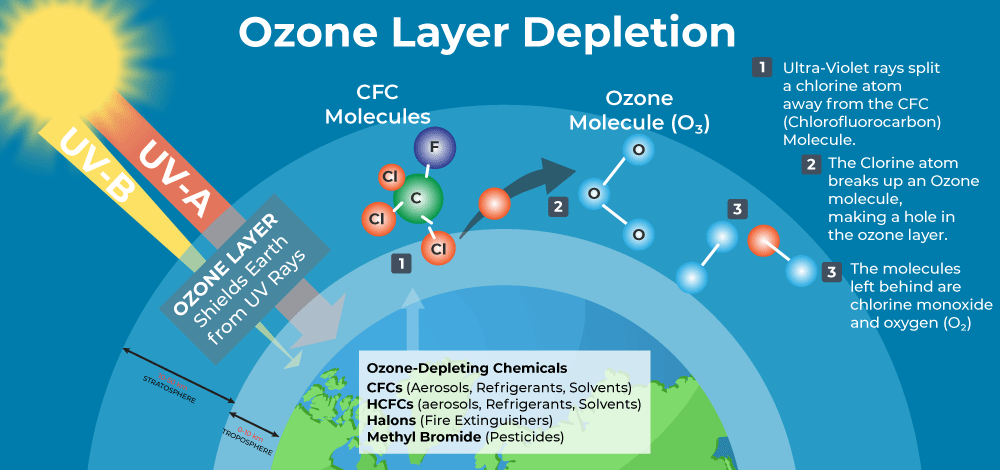
Causes of Ozone Layer Depletion
Ozone layer depletion is a prime concern and ozone layer depletion is caused by multiple factors. The different factors that cause ozone layer depletion are listed below:
Chlorofluorocarbon
The primary factor of ozone depletion and ozone opening is chlorofluorocarbons also known as CFCs, particularly produced by halocarbon refrigerants, solvents, aerosols, sprays, AC, and froth-blowing specialists. CFCs released by them travel to the atmosphere, and their UV rays break the CFC molecule and Chlorine is released. Chlorine reacts with the ozone layer molecule and starts reacting with them.
Nitrogenous Compounds
Different nitrogen compounds like NO2, NO, and N2O also cause ozone layer depletion.
Unregulated Rocket Launches
Rocket launches are also the major cause of ozone layer depletion. Research shows that this is the major factor that causes ozone layer depletion more than chlorofluorocarbon.
Natural Causes
Due to sun spots or stratospheric wind also cause ozone layer depletion. But natural causes are not the prime cause of ozone depletion.
Ozone Depleting Substances (ODS)
Ozone-depleting substances are chemicals that affect or cause ozone layer depletion. For example-Halons, Chlorofluorocarbons, hydrofluorocarbons, etc.
Following are the Ozone depleting substances which cause ozone layer depletion:
| Halons |
Fire-extinguishers |
| Hydrofluorocarbon |
AC, Solvents, Fire-extinguishers. |
| Carbon tetrachloride |
Fire extinguishers, and Solvents |
| Chlorofluorocarbons |
Refrigerators, AC, Solvents, etc. |
| Methyl Chloroform |
Aerosols, and Adhesives |
Effects of Ozone Layer Depletion
Ozone layer depletion directly affect all living organisms. Due to ozone layer depletion, the UV rays direct come to the earth’s surface and do harmful effects on mankind and the environment:
Increased UV Radiation
While ozone is a tiny part of the Earth’s atmosphere, it is responsible for the majority of UVB radiation absorption. The amount of UVB light that penetrates the ozone layer diminishes rapidly with slant-path thickness and density. When stratospheric ozone levels fall, more UVB reaches the Earth’s surface.
Increased Tropospheric Ozone
Increased tropospheric ozone is caused by increased surface UV. Ground-level ozone is widely known as a health danger since ozone is hazardous due to its high oxidant characteristics. Young children, the elderly, and those suffering from asthma or other respiratory problems are especially vulnerable. At the moment, ozone at ground level is mostly formed by the impact of UV radiation on combustion gases emitted by vehicles.
Effects on Crops
An increase in UV radiation is expected to have an impact on crops. A number of commercially significant plant species, such as rice, rely on cyanobacteria living on their roots to retain nitrogen. Cyanobacteria are UV-sensitive and would be harmed by an increase in UV light.
Effect on Humans
Due to ozone depletion UV rays do not absorb and they came directly to the earth’s surface. Humans directly came in contact with UV rays which cause skin cancer, Skin disease, UV rays affect eye cataracts, and mutation in the genome.
Effects on Animals
If animals come in direct contact with UV rays that leads to skin cancer and eye cancer.
Effect on Environment
When plants came in direct contact with UV rays it affects the photosynthesis rate, growth, and flowering of a plant. The whole first also effect if it gets direct exposure to UV rays.
Effect on Marine Ecosystem
Planktons are the most abundant food present in the aquatic ecosystem food chain. UV rays directly affect the plankton which affects the aquatic food chain directly.
Prevention for Ozone Layer Depletion
Ozone depletion affects living organisms in a harmful way. There are some preventive measures to control ozone layer depletion. Such methods are:
Minimize the use of vehicles
Vehicles cause air pollution: release greenhouse gases which are a major factor in global warming and also lead to ozone depletion. Minimizing the use of Vehicles helps to control ozone depletion.
Also read: Greenhouse Effect
Stop using ODS
Avoiding using appliances that release ozone-depleting substances such as AC, refrigerators, etc. Replacing the fire extinguishers with halons-free extinguishers.
Prohibition of Nitrous Oxide
Nitrous Oxide causes ozone depletion. Avoiding Nitrous oxide reaction help to prevent Ozone layer depletion.
Use Eco-friendly Cleaning Products
Most cleaning products contain chemicals that released chlorine and bromine. They find their way into the atmosphere and cause ozone layer depletion.
Also Read:
FAQ’s – Ozone Layer Depletion
1. What is Ozone Depletion and its Effects?
Ozone depletion is the slow loss of ozone in the upper atmosphere as a result of human activity. The Earth’s surface receives more UVB radiation when ozone layer thickness decreases. UVB induces non-melanoma skin cancer and is a significant factor in the growth of malignant melanoma, according to laboratory and epidemiological research.
2. Where does ozone depletion occur?
Stratospheric ozone depletion happens in both hemispheres of the Earth. This is more apparent in the Southern Hemisphere (Antarctica) than in the Northern Hemisphere (Arctic). This is because the creation of the ozone hole is closely related to the temperature of the stratosphere.
3. What are 3 harmful effects of ozone depletion?
Increased UV radiation reaching the Earth as a result of ozone depletion can lead to a rise in skin cancer, cataracts, and compromised immune systems. Too much UV exposure is thought to be contributing to the rise in melanoma, the deadliest of all skin cancers.
4. What causes ozone depletion?
Chlorine and bromine atoms in the stratosphere react with ozone molecules to destroy them. One chlorine atom has the ability to destroy more than 100,000 ozone molecules before it is removed from the stratosphere. Ozone can be destroyed faster than it is normally produced.
5. What is the importance of ozone layer?
The ultraviolet light (UV-B) from the sun is absorbed by the ozone layer between 97% and 99%. This is essential for protecting life on Earth’s surface from dangerous levels of radiation that can disrupt and damage DNA.
Share your thoughts in the comments
Please Login to comment...