A Polymer is a very big molecule or complexly structured material made up of macromolecules, which are small, simple compounds that repeat themselves to make polymers. Both Synthetic Polymers as well as Natural Polymers play significant and relevant roles in daily life. As natural polymers, i.e., DNA and RNA, is the reason for life on earth, without DNA we can’t imagine life on earth.
From well-known synthetic plastics like polystyrene to natural biopolymers like DNA and proteins that are essential to biological structure and function, polymers come in many shapes and sizes. Here are some examples of polymers in the figure given below.
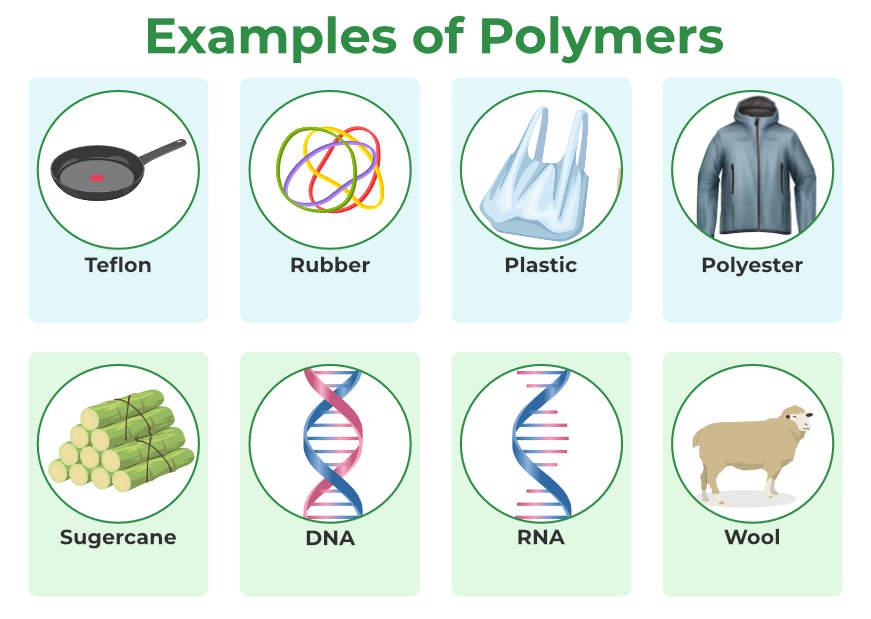
Table of Contents
What are Polymers?
The term “polymer” refers to an unknown amount of monomer units. When there are a lot of monomers, the mixture is sometimes referred to as a high polymer. Monomers of the same molecular weight, shape or chemical makeup are not required for polymers to form. One type of monomer makes up some natural polymers. However, the majority of polymers, both natural and manmade, are composed of two or more different kinds of monomers; these polymers are referred to as copolymers.
Polymer Definition:
A polymer is a molecule made up of numerous small molecules called monomers that are joined together to form a larger molecule. The word “polymer” is made up of two parts: “poly” which means “many,” and “mer” which means “unit.”
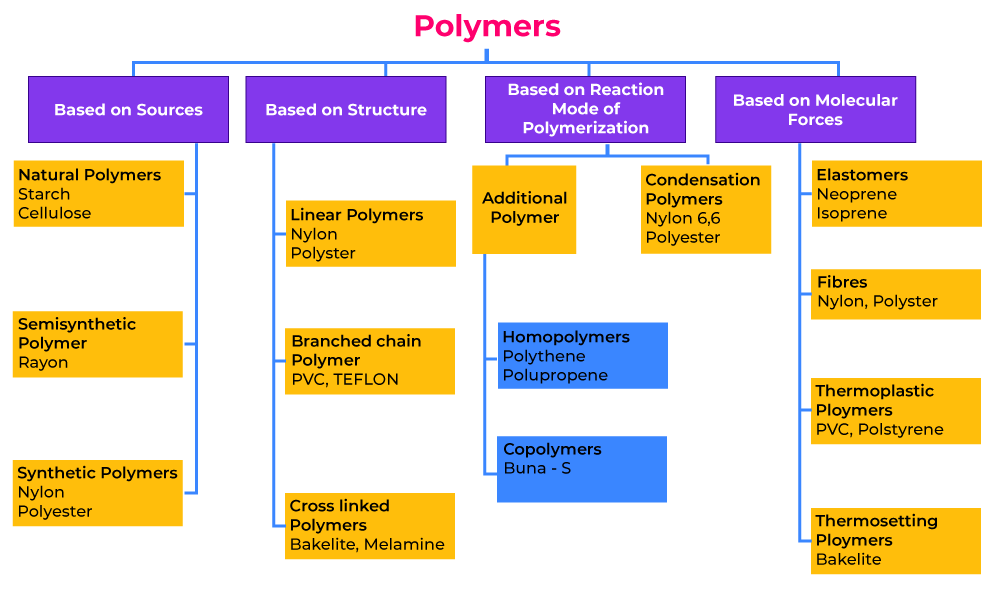
Classification of Polymers
The polymers are classified into various types based on different categories that are as follows:
Classification of Polymers Based on the Source of Availability
On the basis of the structure, polymers can be classified into three categories, which are as follows:
Classification of Polymers Based on Structure
On the basis of the Source of occurrence, polymers can be classified into three categories, which are as follows:
Classification of Polymers Based on Polymerization
On the basis of the polymerization reaction used to form polymers, polymers can be classified into two categories, which are as follow:
Classification of Polymers Based on Monomers
On the basis of monomers used to create polymers, polymers can be classified into two categories, which are as follows:
Classification based on Molecular Forces
Based on the molecular forces between the constituent atom of polymer, polymers can be classified into two categories, which are as follows:
- Elastomers
- Fibers
- Thermoplastics
- Thermosetting Polymers
The Classification of Polymers is illustrated in the chart below:
Structure of Polymers
A hydrocarbon backbone makes up the majority of the polymers in our environment. Due to the tetravalent nature of carbon, a hydrocarbon backbone is a lengthy chain of linked carbon and hydrogen atoms.
The hydrocarbon backbone polymers polypropylene, polybutylene, and polystyrene are a few examples. There are also polymers whose backbones contain other elements in place of carbon. For instance, the backbone of the repeating unit in nylon comprises nitrogen atoms.
Characteristics of Polymers
Depending on the use polymers can be made according to the desired properties and characteristics. Such as:
- Elasticity – Both natural and synthetic polymers exhibit elastic properties which make them useful for Car Tyres, etc.
- Impact Resistant – Polymers can withstand rough handling and harsh usage therefore, would be able to use for luggage, protective cases, etc.
- Tensile Strength – Polymers have high tensile Strength and so are used in making wires, etc.
- Brittle – Polymers are highly brittle, therefore can be deformed easily using heat.
- Reflectivity and Translucence – Some Polymers can be see-through and reflective in nature, which can be best used as reflective film, etc.
Some Polymers and Their Monomers
A monomer is a kind of molecule with the ability to form long chains of chemical bonds with other molecules; a polymer is a chain made up of any number of monomers. Here is the list of Polymers with their monomers.
- Lipids – Here polymers are diglycerides and triglycerides; monomers are glycerol and fatty acids.
- Plastics – In this case, polymers are Polyvinyl chloride (PVC) while monomers are Styrene.
- Proteins – In these polymers are polypeptides; monomers are amino acids.
- Nucleic Acids – Polymers are DNA and RNA; monomers are nucleotides.
- Carbohydrates – Here polymers are polysaccharides and disaccharides while monomers are monosaccharides.
|

|
(a) Phenol (b) Formaldehyde
|
|

|
(a) 1, 3-butadiene (b) Vinyl Cyanide
|
|
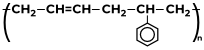
|
(a) 1, 3-butadiene (b) Styrene
|
|

|
(a) Ethylene glycol (b) Phthalic acid
|
|
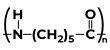
|
Caprolactam
|
|
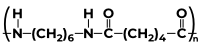
|
(a)Hexamethylene Diamine (b) Adipic Acid
|
|
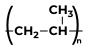
|
Propene
|
|
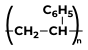
|
Styrene
|
|
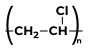
|
Vinyl Cyanide
|
|

|
Isoprene (1, 2-methyl 1 – 1, 3-butadiene)
|
|

|
Tetra Fluoro Ethane
|
|
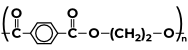
|
(a) Ethylene glycol (b) Terephthalic acid
|
How are Polymers Made? – Polymerization Reactions
The chemical reaction involved to produce a Polymer is called Polymerization Reaction.

The method of producing polymers is called polymerization. Following processing, these polymers are used to create a variety of plastic items. Smaller molecules, or monomers, are joined chemically to form larger molecules, or macromolecules, during polymerization. A polymer is created by hundreds of these macromolecules working together.
The molecules that make up the monomer could all be the same or could stand in for two, three, or more different substances. In order to create a product with specific physical characteristics that set polymers apart from substances made of smaller and simpler molecules, such as elasticity, high tensile strength, or the capacity to form fibers, typically at least 100 monomer molecules must be combined; frequently, thousands of monomer units are incorporated into a single polymer molecule.
Types of Polymerization Reactions
There are two types of polymerization reactions, which are as follows:
Addition Polymerization
In Addition Polymerization, small monomer units join together to form a giant polymer molecule. In each step of polymerization of this kind, the length of the polymer chain increases, that’s why it is also called Chain-Reaction or Chain-Growth Polymerization.
For example, the formation of polyethylene from ethylene, the formation of PVC, and the formation of Teflon are some examples of the addition polymerizations.
Condensation Polymerization
In Condensation Polymerization, monomers including bifunctional groups such as diamines, -idols, dials, dicarboxylic acid, etc. undergo polymerization, and water, CO, and ammonia small molecules got eliminated to form polymers. The formation of nylon -6,6 is an example of Condensation Polymerization.
Also Check: Step Growth or Condensation Polymerization and Types of Polymerization
How to Calculate the Molecular Mass of Polymers?
The total atomic weight of all the molecules that make up a polymer is known as the Molecular Weight of the Polymer.
A polymer is made up of repeating units. The monomer molecules were originally these repeating units. Polymer chains have different weights and lengths when they first form. The ability to describe the polymer structure is important. Any characterization of a polymer essential is either the weight-average or number-average molecular weight.
Number Average Molecular Mass (Mn)
Number mean or simple arithmetic mean, represents the weight of all molecules divided by the number of molecules.
Mathematically, this can be calculated as:
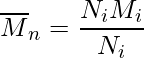
where,
- Ni is the average number of molecules, and
- Mi is the average molecular mass.
Weight Average Molecular Mass (Mw)
Weight average or simple arithmetic mean, represents the mass and molecular masses of all molecules divided by mass.
Mathematically, this can be calculated as:
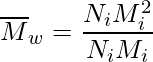
Degree of Polymerization
The degree of Polymerization represents the number of monomeric units in a Polymer. It is defined as the ratio of the molar mass of polymer to the molar mass of the monomeric unit and is given as,
DP or p = M / n
where,
- M is the molar mass of the polymer, and
- n is the molar mass of the monomeric unit.
Uses of Polymers
Polymers have a wide range of applications, such as:
- Polypropylene has a wide range of uses, including in plastics, textiles, packaging, stationery, and airplanes.
- Polystyrene, one of the most used plastics, is frequently used in packaging. Products made of polystyrene include toys, trays, plates, cabinets for televisions, bottles, and lids. It can therefore also be utilized as an electrical insulator.
- PVC is a polymer that is mostly used to create sewage pipes. This material is also used to make cable insulators.
- Polyvinyl chloride is highly used in the manufacturing of doors and windows in addition to being used in clothing and furnishings.
- Due to the high molecular mass of these polymers, the urea-formaldehyde resin can be used to create a wide variety of products. Products including adhesives, molds, laminated sheets, and others are included in this.
Also Check: Polymers of Commercial Importance
FAQs on Polymers
Q1: Define vulcanization of rubber.
Answer:
Natural rubber has a low physical stability due to its strong elasticity. Sulfur at a concentration of 5% promotes the crosslinking of the linear chains, stiffening the rubber for use in tyre applications.
Q2: What are examples of biodegradable polymers?
Answer:
The examples of biodegradable polymers are Poly-β-hydroxybutyrate-co-β-hydroxy valerate (PHBV), Polyglycolic acid (PGA), Polylactic acid (PLA), Poly (ε-caprolactone) (PCL) and Nylon-2-Nylon-6.
Q3: What is Engineering Plastics?
Answer:
The polymers with high-performance synthetic resin, high durability and heat resistance are called the Engineering Plastics. Examples: Silicone, Polycarbonate, ABS, and Polysulfone.
Q4: What are Polymers used for?
Answer:
Polymers are used for various purposes like, manufacturing synthetic fibers, disposable crockery, fiberglass, plastic paints, teflon-coated cookware, etc.
Q5: Write some physical Properties of Polymers.
Answer:
Here are some important physical properties of polymers –
- Both natural and synthetic polymers exhibit elastic properties which make them useful for Car Tyres, etc.
- Polymers can withstand rough handling and harsh usage therefore, would be able to use for luggage, protective cases, etc.
- Polymers has high tensile Strength and so are used in making wires, etc.
- Polymers are highly brittle, therefore can be deform easily using heat.
- Some Polymers can be see-through and reflective in nature, which can be best use as reflective film, etc.
Q6: What are Biodegradable Polymers?
Answer:
These polymers break down after their purpose by bacterial decomposition process to result in natural byproducts. For example: Poly β-hydroxybutyrate -co-β-hydroxy valerate (PHBV).
Share your thoughts in the comments
Please Login to comment...