Classification of Amines
Last Updated :
15 Mar, 2022
Amines are an important class of organic compounds. They are widely found in both plants and animals. They are found among proteins, vitamins, alkaloids, hormones, etc. Synthetic examples include polymers, drugs, dyestuff, etc. These amines are widely used. For example, quinine is an important anti-malarial drug, adrenaline and ephedrine are used to raise blood pressure, Novocaine is used as an anesthetic in dentistry, codeine is used as an analgesic (as a pain reliever). Benadryl is used as an antihistamine drug. Quaternary ammonium salts are used as surfactants.
Amines are considered to be derivatives of ammonia in which one, two, or all three hydrogen atoms are replaced by an alkyl or aryl group.
Classification of Amines
Amines are broken down as primary (1°), secondary (2°), or tertiary (3°) because one, two, or three hydrogens of the ammonia molecule are displaced by alkyl or aryl groups in the ammonia molecule. However, we get RNH, or ArNH2 if one hydrogen atom of ammonia is replaced by an alkyl (R) or aryl group (Ar). Nevertheless, we take R-NH-R, which is the 2° amine, if the two H atoms of the ammonia H atom of RNH are replaced by another alkyl (R) or Ar group. And the alkyl or aryl groups may be the same or different.
Displacement of all three H atoms of ammonia or the other H of R-NH-R by an alkyl (prime high) or aryl group gives Rn(prime) prime which can be 3° amines (R”) equal or unequal. R or R’).
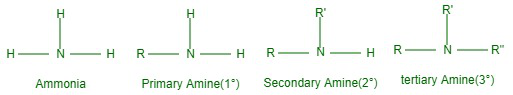
The characteristic groups in primary, secondary, and tertiary amines are amino, imino, and tert- nitrogen.
In addition to these three types of amines, there is another class of compounds known as quaternary ammonium pounds. These compounds can be thought of as derivatives of ammonium salts in which all four H-atoms are held by alkyl or aryl groups. for example, Tetramethylammonium iodide, Tetramethylammonium Chloride, TetraTetramethylammoniummethyl ammonium hydroxide, Tetra methyl ammonium bromide, etc.
Amines can be further divided into two categories:
- Aliphatic Amines: Amines in which the nitrogen atom is directly bonded to one or more alkyl groups are called aliphatic amines. For example, methylamine (1°), dimethylamine (2°), and trimethylamine (3°).
- Arylalkyl amines or side-chain substituted amines: Amines in which the nitrogen atom is attached to a side chain of an aromatic ring are called alkylamines. for example, Benzylamine (1°), dibenzylamine (2°) and tribenzylamine (3°).
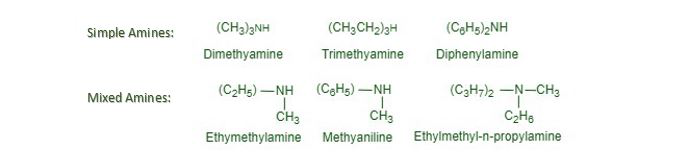
Simple and mixed amines. Secondary and tertiary amines may be classified as simple or mixed amines according to as all the alkyl or aryl groups attached to the nitrogen atoms are the same or different. For example,
Physical Properties of Amines
- Solubility: Aliphatic amines (1°, 2°, and 3°) form H-bonds with water and are therefore soluble in water. However, aromatic amines are insoluble in water due in large part to hydrocarbons.
- Boiling points- The 1° amine has a higher boiling point than the 3° amine due to the presence of two H-atoms directly attached to N, resulting in a higher H-bonding in the 1° amine.
Basic Character of Amines
- Aliphatic Amines
- All aliphatic amines are more basic than ammonia.
- In aqueous solutions, methylamine follows the basicity order (CH₂)₂NH > CH₂NH₂ > (CH₂), N (fe 2° 1 > 3º), but for ethylamine and all other higher amines, the basicity order is R₂NH > R₂N > RNH. follows. (ie 2° > 3° > 1°).
- The order of basicity in the gaseous state is 3° > 2° > 1º amines.
- The higher the value of Kp or the smaller the value of pKb the stronger the base.
- Aromatic Amines
- All aromatic amines are weaker bases than ammonia.
- Electron donating groups like -CH3, – OCH3, -NH2 increase basicity while electron-withdrawing substances like -NO2, -CN, -(halogens) decrease the basicity of amine. The effect of these substances is more pronounced at p- than at m- conditions.
- o-substituted anilines are weaker bases than anilines, regardless of the electron-donating or electron-withdrawing nature of the substituents. This is called the ortho effect and is caused by steric hindrance.
Aliphatic amines are more basic than aromatic amines. Like aromatic amines, the lone pair of electrons present on nitrogen participates in resonance and is therefore not available for donation, whereas it is available in aliphatic amines.
Sample Questions
Question 1: Give a chemical test to differentiate between ethylamine and aniline.
Answer:
Ethylamine is a primary aliphatic amine whereas aniline is a primary aromatic amine. Both can be identified by the azo dye test. When aniline is treated with HNO, (NaNO₂+ dil. HCl) at 273K, followed by treatment with an alkaline solution of 2-naphthol, the orange color is obtained, but due to the formation of ethylamine. With N2 gas gives strong effervescence of 1 alcohol.
Question 2: Give a chemical test to differentiate between aniline and N-methylamine.
Answer:
Aniline is a primary aromatic amine while N-methyl aniline is a secondary aromatic amine. The two can be separated by the carbylamine reaction. Upon mixing CHCl3 and KOH, aniline will give off an offensive odor due to the formation of phenyl carbylamine (phenyl isocyanide), while N-methyl aniline will not react.
Question 3: Why does electrophilic substitution occur more readily in aromatic amines than in benzene?
Answer:
Due to the strong activating group -NH₂, the electrons on the N-atom in aniline are denoted above the benzene ring. As a result, the electron density on the benzene ring increases as compared to benzene. Therefore, aniline becomes activated and electrophilic substitution occurs more easily in aniline.
Question 4: Give a chemical test to differentiate between aniline and benzylamine.
Answer:
Aniline and benzylamine can be separated by azo dye test. Aniline reacts with NO2 at 273-278 K to form stable benzene diazonium chloride which gives orange color on reaction with an alkaline solution of β-naphthol. Whereas benzylamine does not give this test.
Question 5: p-methoxy aniline is a stronger base than aniline but p-nitro aniline is a weaker base than amines. Explain.
Answer:
The methoxy group (-OCH3) is the electron freeing group and increases the electron density on the N atom. Therefore, it has a tendency to donate more electrons than aniline and thus is a stronger base than aniline. On the other hand, the nitro group is the electron withdrawing group and hence, the electron density on the nitrogen atom decreases. Consequently, p-nitroaniline is a weaker base than aniline.
Question 6: Can we prepare aniline by Gabriel’s phthalimide reaction?
Answer:
The Gabriel phthalimide reaction to aniline cannot be made because it requires treatment of potassium phthalimide with C6H5Cl or C6H5Br. Since aryl halides do not undergo nucleophilic substitution reactions under normal conditions, the reaction does not occur. Therefore, aniline cannot be prepared by this method.
Like Article
Suggest improvement
Share your thoughts in the comments
Please Login to comment...