Pure and Impure Substances
Last Updated :
14 Jul, 2021
Substance is a matter which is associated with a set of definite properties and composition. Every pure compound and element is a substance. Substances are the materials composed of atoms and molecules. Every substance is recognized by a specific weight and volume. It cannot be segregated into other different types of matter by performing any physical process.
Examples:
- Silver
- Salt
- Metals, non-metals
Pure Substances
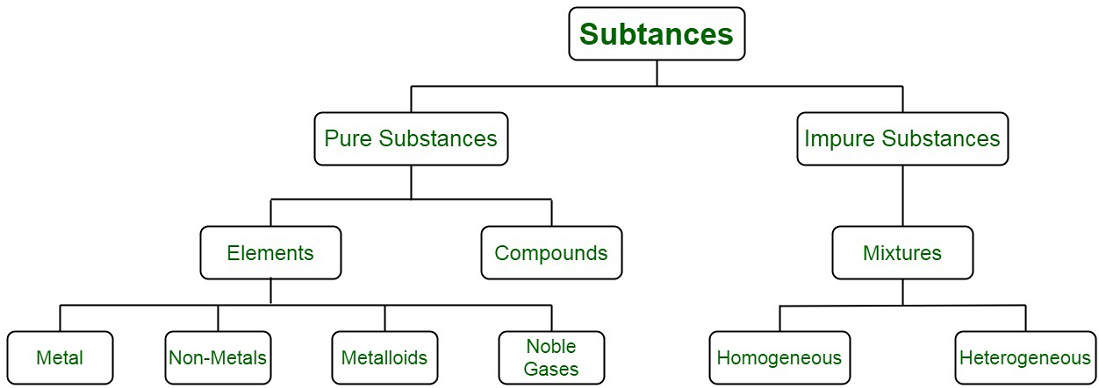
Pure substances are composed of a single or atomic type of particles. They possess a constant structure. Pure substances can be further categorized into elements and compounds respectively:
An element is considered to be a pure substance consisting of singular type of atoms. It can neither be broken down nor transformed into new substances by any kind of processes, be physical or chemical process. Elements are mostly categorized into metals, non-metals or metalloids. For instance, gold and silver are elements.
A compound is a pure substance where two or more elements are combined using chemical means in a fixed defined ratio. These substances can be broken down and transformed into new substances by chemical methods. For instance, carbon dioxide.
Properties of pure substances
Pure substances have the following properties:
- Almost all are homogeneous in nature.
- Possess fixed melting and boiling points.
- Uniform composition.
Examples
- Sugar
- Baking Soda
- Oxygen
- Ammonia
- Water
- Protein
- Diamond
- Chlorine

Impure Substances
Impure substances, also termed as mixtures are composed of atoms or molecules belonging to different types. They do not possess constant or fixed structure throughout. Impure substances can be converted to pure ones by using different separation techniques. For instance, a mixture of naphthalene balls and common salt can be separated by performing sublimation. Sublimation results in the extraction of common salt, since the moth balls directly pass into the vapour state.
Mixtures can be categorized into homogeneous and heterogeneous mixtures. Homogeneous mixtures have a uniform composition throughout the volume of the substance whereas the heterogeneous mixture contains a varying composition.
Properties of Impure Substances
Impure substances have the following properties :
- The properties of the resulting mixture is an average of the properties of the constituents.
- Formed as a result of physical change.
- May be either homogeneous or heterogeneous.
- Substances have a variable composition.
Examples:
- Rocks
- Muddy water
- Salad
- Sand in water
- Salt in water
- Vegetable soup

Differences between Pure and Impure Substances
|
Pure Substances
|
Impure Substances
|
| Made of a single element or compound |
Several elements & compounds together form an impure substance. |
| They are categorised into solid, liquid, gases. |
Categorised as Heterogeneous and homogeneous. |
| Physical properties are constant. |
Physical properties are varying. |
| Purity is 100% |
Purity is less than 100%. |
| Chemical properties are constant. |
Chemical properties are varying. |
| Example: Gold, pure water, hydrogen gas |
Example: Oil & water, sand & sugar |
Sample Questions
Question 1: Why is pure oxygen gas termed as an element?
Answer:
The pure oxygen gas belongs to the category of element, though it consists of molecules. This is because the molecules forming the gas all belong to a single type of element, whereas compounds are composed of one or more different types of elements.
Question 2: Define metals giving examples.
Answer:
Metals belong to the category of elements which are identified by various properties lustre, ductile ,malleable and are mostly good conductor of heat and electricity. Example – iron, manganese.
Question 3: Differentiate between homogeneous and heterogeneous mixtures.
Answer:
|
Homogeneous Mixtures
|
Heterogeneous Mixtures
|
| Uniform composition throughout the mixture |
Non-Uniform composition throughout the mixture |
| Components are not visible to the naked eye. |
Components are visible to the naked eye. |
| The entire mixture is in the same phase. |
The entire mixture is not in the same phase. |
| Particle size is estimated at atomic or molecular level |
Large particle sizes. |
| Components cannot be easily segregated. |
Components can be easily segregated. |
Question 4: Why is salt water not considered as a substance?
Answer:
Different samples of salt water may contain different quantities of salt in the water used. Therefore, different samples don’t have a uniform chemical composition throughout.
Question 5: Can fresh orange juice be considered pure substance?
Answer:
Orange juice may contain pulp, which acts as a solute in this case, and liquid. It doesn’t have a uniform chemical composition throughout, therefore, it is not considered to be a pure substance.
Question 6: How can we recognise pure substances?
Answer:
If we can write a chemical formula for the substance being provided then it is a pure substance. For example : aluminium oxide.
Question 7: Give an example of impure substance from daily life.
Answer:
Ghee is an impure substance, composed of various constituents like amino acids and fatty acids and salts.
Share your thoughts in the comments
Please Login to comment...