Uses of Metals and Non-Metals
Last Updated :
23 Feb, 2022
There is matter all around us in nature and the most amazing thing to notice is that not all matter act or respond the same way if they are put under similar conditions, this is due to the fact that matter is made up of basic component known as an element, an element is the most basic and purest form of any matter and the matter is made of up these elements. Matter can either be present in its pure form when only one type of element is used to form the matter, or it can be the combination of two types of elements. Elements are not all same types, but they are of three different types, this is the reason why they react differently under similar conditions. Let’s learn in more detail about the types of Elements.
Elements
Elements are substances that can not be broken into smaller substances further, they are already present in their purest form without any adulteration of impurity. Elements are the fundamental material that is combined to form matter. For example, Iron, Helium, Oxygen, Sulphur, etc.
Elements can be divided into two types:
Metals
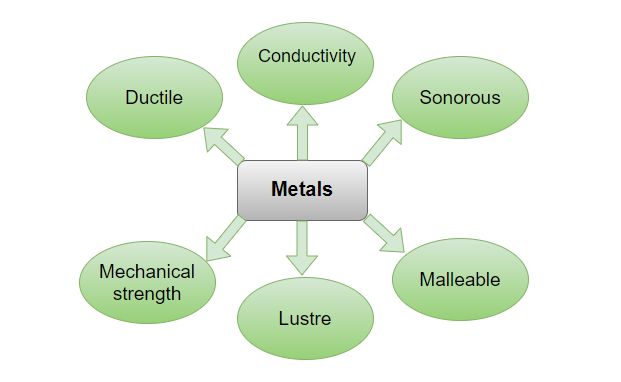
Metals are described as those elements that lose valence electrons in order to create a positive charge, known as cations. They are the reason for the conductivity of metals. Metals are described as those substances that have the properties like malleability, ductility, sonority, lustre, conductivity, and they are mostly solid in nature. Some Examples of metals are, Iron, Gold, Silver, Aluminium, Copper, etc.
Properties of Metals
Physical Properties
- Thin sheets of metal can be hammered out of metals. It signifies they have the ability to be moulded.
- Metals have a ductile property. Wires can be made out of them.
- Metals are excellent heat and electrical conductors.
- Metals are lustrous, meaning they have a gleaming finish.
- Tensile strength is a property of metals. It suggests they’re capable of supporting heavyweights.
- Metals have a resonant quality to them. It means they generate a ringing sound when we strike them.
- Metals are abrasive. It means they are difficult to cut.
Chemical Properties
- Reaction with Oxygen: Metals react with oxygen to generate metallic oxides, which are naturally basic. Magnesium, for example, is a metal that interacts with oxygen to generate magnesium oxide. Magnesium hydroxide is formed when magnesium oxide is dissolved in water. The reactions are given below.
2Mg + O2 -> 2MgO
MgO + H2O -> Mg(OH)2
- An aqueous solution of metal oxide transforms red litmus paper into blue litmus paper.
- Water Reaction: Metals react with water. Metal hydroxide and hydrogen gas are generated when a metal reacts with water. As an example, when sodium metal combines with water, sodium hydroxide and hydrogen gas are formed. The response is presented below.
Na + H2O -> NaOH + H2
- Metal Reaction with Acid – Metals react with acids, forming salt and hydrogen gas. Some metals react violently with acids. Magnesium metal, for example, interacts with hydrochloric acid to generate magnesium chloride and hydrogen. The reaction is as follows –
Mg + 2HCl -> MgCl2 + H2
- Metals react with bases, such as sodium hydroxide, to generate salt and hydrogen gas. For example, when aluminium metal interacts with sodium hydroxide, sodium aluminate and hydrogen gas are formed. The reaction is as follows –
Al + NaOH -> NaAlO2 + H2
- A more reactive metal displaces a less reactive metal from its salt solution in a displacement reaction. Displacement reactions are the name given to these types of reactions. Iron, for example, is more reactive than copper. As a result, it displaces copper from its salt. The response is presented below.
CuSO4 + Fe -> FeSO4 + Cu
Uses Of Metals
- Metals are used to make electric wires because of their conductivity and low resistance. Some good examples of such metals are copper and aluminium.
- In Haber’s process, Iron is used as a Catalyst. In shorts, some metals are used as a catalyst in chemical reactions in order to fasten the process.
- In order for proper heat transfer, some metals are used as household utensils or factory equipment. The example of such metals are Iron, aluminium, and copper.
- In order to protect Iron from rusting, which is really common in iron, it gets rusted in the presence of oxygen and moisture, Zinc is used to protect it from rusting. The process of applying the Zinc layer is known as the Galvanization process.
- One very important and dangerous metal that is seen in our medical kit is mercury which is used in thermometers.
- Car batteries are made up of Lead which is a metal.
- Some rarely available metals like Gold and silver are used to make ornaments, sometimes, very thin sheets of gold and silver are used to keep on top of sweets.
- Chromium and nickel are used in stainless steel.
- Chromium and nickel are also used in the process known as Electroplating.
- In the making of bullet-proof steels, Zirconium is used.
- Since metals are ductile in nature, aluminium is very famous for forming very thin sheets. It is used for packaging purposes.
Non-Metals
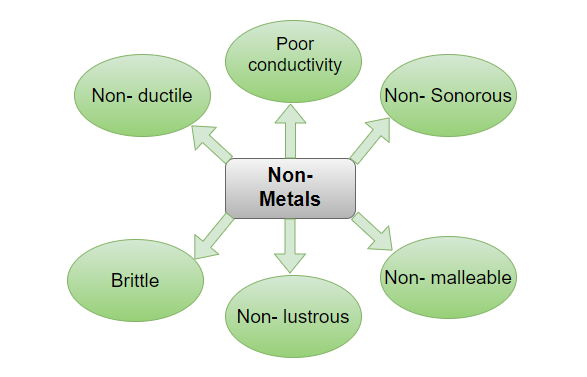
Non-metals are those elements that are not able to produce electricity or have conductance nor do they produce heat, these elements are the opposite of metals. The properties of non-metals include non-lustrousness, non-sonorous, non-ductile, non-malleable and poor conductor of heat and electricity. They are structurally brittle in nature and can be used in rolling, pressing, moulding, etc. Some examples of non-metals are Carbon, Oxygen, Nitrogen, Arsenic, etc.
Properties of Non-metals
Physical Properties
- Nonmetals are unable to be hammered into thin sheets. It signifies they lack the property of malleability. Instead, they are brittle and split up into pieces when hammered.
- Nonmetals have no ductility. They are unable to be attracted into wires.
- Nonmetals are poor heat and electrical conductors.
- Non-metals are not lustrous, meaning they are not shiny. Rather than being bland in appearance.
- Tensile strength is low in nonmetals. This means they can’t support heavyweights.
- Non-metals have no sonorous properties. It means that when we strike them, they produce no sound.
- Nonmetals are pliable. It suggests they can be easily cut with a knife.
Uses of Non-metals
- Hydrogen is a very special element and also a non-metal that is used for so many different purposes.
- In order to convert Vegetable oil into vegetable ghee, the process required is Hydrogenation and hydrogen is required in the process.
- Hydrogen is also used in the manufacturing of Ammonia which is used for different purposes.
- In order to make rocket fuel, liquid hydrogen is used.
- For the Vulcanization of Rubber, Sulphur is used.
- Sulphur is also used to make gunpowder.
- In order to manufacture Sulphuric acid (H2SO4), sulphur is required.
- Nitrogen is used to preserve Food materials, for example, the bag of chips is kept for days and still does not go bad, it is due to the presence of nitrogen inside the packet.
- Components of Nitrogen, such as, TnT (Tri Nitro Toluene) is used to make explosives.
- Nitrogen is also used in the making of fertilizers, ammonia, nitric acid.
- Carbon is used to make electrodes and dry cells that are used in batteries.
Conceptual Questions
Question 1: Which metal is the best conductor of electricity?
Answer:
Silver is the best conductor of heat and electricity.
Question 2: Which Element is used in the instrument which is used to measure temperature? State one distinguishing property of the elements.
Answer:
Mercury is used in the instrument (thermometer) which is used to measure temperature. Mercury has one different property as a non- metal, that is, it is naturally present in liquid state.
Question 3: Why Silver is not used in making electric wires even after being the best conductor of electricity?
Answer:
Silver is not used as electric wires even after being the best conductor of electricity because silver is not present in abundance on earth, which makes it one of the precious metals found on earth. Using silver as electric wires will be very costly and the wires can be easily stolen as well.
Question 4: Iron is a metal that is also present in our bodies. Where exactly is iron found in our bodies?
Answer:
Iron is necessary for human body, it is present in the Red blood cells, the red pigment of the red blood cells is due to the presence of iron, it is known as Hemoglobin.
Question 5: Which element is used for preserving food? Is it a metal or a non-metal?
Answer:
The element used for preserving food items is Nitrogen. Nitrogen is a non-metal.
Like Article
Suggest improvement
Share your thoughts in the comments
Please Login to comment...