Diamagnetic Materials – Definition, Properties, Applications
Last Updated :
03 Sep, 2021
The genesis of magnetism is due to the spin motion of electrons and their interactions with one another. Describing how materials respond to magnetism is the greatest approach to present different sorts of magnetic materials. You might be surprised to learn that all matter is magnetic. The main distinction is that certain materials have a higher magnetic permeability than others. What separates them is the degree of interaction between the magnetic moments. A few materials have no collective atomic magnetic moment interaction, whereas others have a high atomic magnetic moment interaction.
Classification of Magnetic Materials
The response of a material to an external magnetic force is defined by its magnetic characteristics. Solids may be divided into three categories based on their magnetic properties:
- Ferromagnetic – materials that are attracted to magnetic materials strongly. Nickel, Cobalt and Iron are a few of its examples.
- Paramagnetic – materials that are attracted to magnetic materials weakly. Molybdenum, Lithium and Magnesium are a few of its examples.
- Diamagnetic – materials that aren’t attracted to magnetic fields. Gold, Water, Mercury, Bismuth and Copper are a few of its examples.
What are Diamagnetic Materials?
When put in a magnetic field, diamagnetic materials are those that are freely magnetised. The magnetization, on the other hand, is in the opposite direction of the magnetic field. Diamagnetism is the term for the magnetism displayed by certain materials.
A magnet generally repels diamagnetic materials. Technically, these solids produce an induced magnetic field in the opposite direction of an externally applied magnetic field and are repelled by it. The behaviour of paramagnetic materials is exactly the reverse of this phenomenon. Magnetic fields are created by the orbital motion of electrons on the atoms of diamagnetic materials, which forms small atomic current loops. When a material is subjected to an external magnetic field, these current loops tend to align in a way that opposes the applied field.
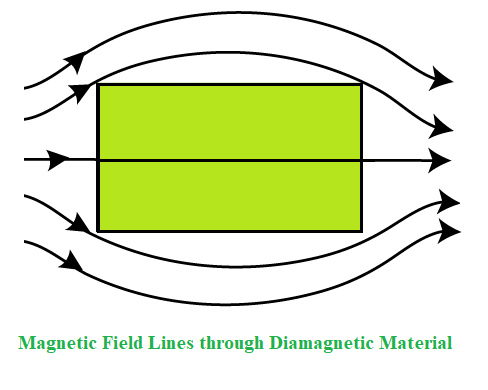
Because all the electrons in diamagnetic materials are coupled, there is no permanent net magnetic moment per atom. Diamagnetic characteristics result from the realignment of electron pathways due to the impact of an external magnetic field. As a result, all materials with paired electrons have diamagnetic characteristics.
Properties of Diamagnetic Materials
- Diamagnetic materials do not have atomic dipoles because the resulting magnetic moment of each atom is zero owing to paired electrons.
- A magnet will resist diamagnetic materials.
- Because the field repels the substances only weakly, they tend to migrate from a strong to a weak region of the external magnetic field in a non-uniform field.
- The magnetization’s strength (I) is a tiny, negative number that is proportional to the magnetic field.
- Magnetic susceptibility is negligible and tiny.
- The permeability ratio is somewhat less than one.
- The temperature does not affect diamagnetic materials. Curie’s law does not apply to these materials.
- When hanging in a homogeneous magnetic field, a rod of diamagnetic material comes to rest with its length perpendicular to the field direction because the field is highest at the poles.
- In a U-Tube, a diamagnetic liquid depresses the Limb between the poles of a magnet.
- The magnetic dipole moment is tiny and polarised in the opposite direction as the magnetic field H.
- If a diamagnetic liquid is placed in a watch glass between two pole pieces that are very near to one other, liquid collects on the sides and depression appear in the centre, where the field is greatest.
- When a liquid is poured on a watch glass that is placed over two pole pieces that are suitably spaced apart (more than in the previous example), liquid collects in the middle, where the field is weakest.
- The induced dipole moment caused by a shift in the orbital motion of electrons in atoms caused by the applied field is the source of diamagnetism.
Application of Diamagnetism Materials or Diamagnetism
- Since diamagnetism is essentially the expulsion of magnetic fields within a material, powerful diamagnetic materials may be levitated, or levitate magnets if they are sufficiently strong and large enough. The diamagnetic levitation of pyrolytic graphite over permanent neodymium magnets is seen in the diagram below.
- The diamagnetic response in superconductors leaves zero internal magnetic fields, as seen in the picture below. The Meissner effect describes how certain materials may be easily levitated in the presence of a powerful permanent magnet, as seen in the diagram above.
- Superconductors with high temperatures (−100 K), on the other hand, are manufactured from unusual materials that need expensive processing and require cryogenic fluids to achieve the superconducting state.
- The superconductor (blue colour) in the figure below expels all of its external magnetic fields, which are referred to as perfect diamagnets.
Important Points To Remember
Diamagnetism exists in all materials and is temperature independent, but because the effect is so mild in contrast to paramagnetism and ferromagnetic effects, it is frequently overlooked.
Gases, liquids, and solids can all exhibit diamagnetism.
Superconductors – Superconductors are essentially diamagnetic materials with a volume susceptibility of χv = − 1(dimensionless). They exhibit complete diamagnetic screening and may be classified as perfect diamagnets since they expel all magnetic fields.
Meissner Effect – When a permanent magnet is introduced close to a superconductor, the superconducting material generates a current that fully opposes the permanent magnet’s magnetic field. The superconductor expels an applied magnetic field, resulting in a field of zero in its interior. In the Meissner state, a superconductor acts as a perfect diamagnet.
Sample Problems
Problem 1: Why is diamagnetic susceptibility negative?
Solution:
Because the electron pairs in diamagnetic materials are all together, there are no total spins. The magnetic field of these materials is directed in the opposite direction as the applied magnetic field. The diamagnetic has a low negative susceptibility, implying that it is diamagnetic.
Problem 2: Is water naturally diamagnetic? Why?
Solution:
Water is diamagnetic, according to popular belief. Only the electron pairs are found in diamagnetic compounds. Water, on the other hand, includes two bonding pairs of electrons between hydrogen and oxygen atoms, as well as two lone pairs of electrons in the oxygen atom. At least one unpaired electron exists in paramagnetic compounds. When a molecule contains an odd number of electrons, something happens (like in NO). It can also happen when a few molecules have an even number of electrons (as in O2). The water repels the magnet, as we can see. This happens because any magnetic field near water produces its own magnetic field, repelling the magnet, a phenomenon known as diamagnetism.
Problem 3: What are the Diamagnetic Materials?
Solution:
Diamagnetic materials resist the magnetic field in the same way as an external magnetic field repels it, but they also create an induced magnetic field in the opposite direction, resulting in a repulsive force.
Problem 4: A diamagnetic substance is heated from 200 to 450 degrees Celsius. What is the diamagnetic susceptibility change?
Answer:
Temperature has no effect on diamagnetic susceptibility, therefore heating a material will not affect its diamagnetic susceptibility.
Problem 5: Give few examples of diamagnetic substances.
Solution:
Copper, Zinc, Bismuth, Silver, Gold, Antimony, Marble, Water, Glass, NACL, and other diamagnetic compounds are only a few examples.
Like Article
Suggest improvement
Share your thoughts in the comments
Please Login to comment...