Organic compounds are defined as chemical compounds which contain carbon atoms linked with other elements through simple covalent bonds. These elements could be connected by single covalent bonds, double covalent bonds, or triple covalent bonds. In other words, we can say that all organic compounds contain carbon as their central atom. We must note that all compounds containing carbon are not considered organic compounds, such as carbonates(-CO3), bicarbonates(-HCO3), etc are carbon-containing compounds but not organic. Whereas we can say with surety that all organic compounds contain carbon atoms but vice versa is not true.
In this article, we will learn about organic compounds and their various classification based on bonds, functional groups, and structure. As there are millions of organic compounds and studying them individually is not possible. Therefore, the classification of organic compounds is required. So, let’s start learning about Organic Compounds and their classification.
Organic Compounds
Organic Compounds are those chemical compounds that are majorly derived from livings beings. The branch of chemistry which deals with the study of organic compounds is called Organic Chemistry. The most significant feature of an Organic Compound is that it contains Carbon atoms but not all compounds that have Carbon atom is organic in nature. For Example, CO and CO2 are not organic in nature. Hence, in order to identify an Organic Compound, we must look that if it contains an open or closed chain of Carbon atoms with hydrogen atoms linked to it or any other atom in place of hydrogen. For Example, CH4 and CCl4 both are organic in nature.
Earlier it was thought that Organic Compounds can only be derived from living things but the major breakthrough happened when Friedrich Wohler in 1828 made the first-ever organic compound in the lab and it was called Urea which has the molecular formula of NH2CONH2. Urea is one of the most important compounds that we use in the agricultural field to fulfill the deficiency of nitrogen in the soil. Not only this, over the period we have developed organic compounds which are helpful and advantageous for industrial purposes and hence mankind. The biodegradable nature, economical feasibility, and easy availability of organic compounds make it important for our daily utilities.
Since there are millions of organic compounds and we can make even more by substituting branched atoms and by simple reactions, it makes us mandatory for us to classify them for our easy understanding.
What is an Organic Compound?
Organic compounds are compounds that are produced by living organisms. They are very useful in all living organisms’ reproduction, development, and working. Organic Compounds have special characteristics, in that they all contain carbon atoms. Carbon has a special property called catenation property which helps it to form long-chained compounds. Carbon has four electrons in its outermost shell so it can form four covalent bonds to complete its octet. Carbon forms covalent bonds with other carbon atoms along with it also form bonds with Hydrogen, Nitrogen, Oxygen, and other atoms.
Examples of Organic Compounds
Some examples of common Organic Compounds are as follows:
- Methane (CH4): A most Basic organic compound.
- Ethanol (C2H5OH): A colourless liquid alcohol used as a solvent, and fuel, and also used in alcoholic beverages.
- Glucose (C6H12O6): A simple sugar that is an essential energy source for living organisms.
- Amino Acids: Amino acids are building blocks of life which form all the proteins necessary for life to exist.
- Chlorophyll (C55H72O5N4Mg): Green pigment in plants which gives plants their green colour and help them make food as well.
- Vitamin C (C6H8O6): Vitamin C is a water-soluble vitamin which is essential for the growth and repair of the human body.
- DNA (Deoxyribonucleic Acid): A complex organic molecule which stores the instruction for life.
- Polymers: Large molecules made with repeating subunits known as monomers. Some examples include polyethene and polystyrene.
Classification of Organic Compounds
A general classification of the organic compound on the basis of structure is described in the below image.
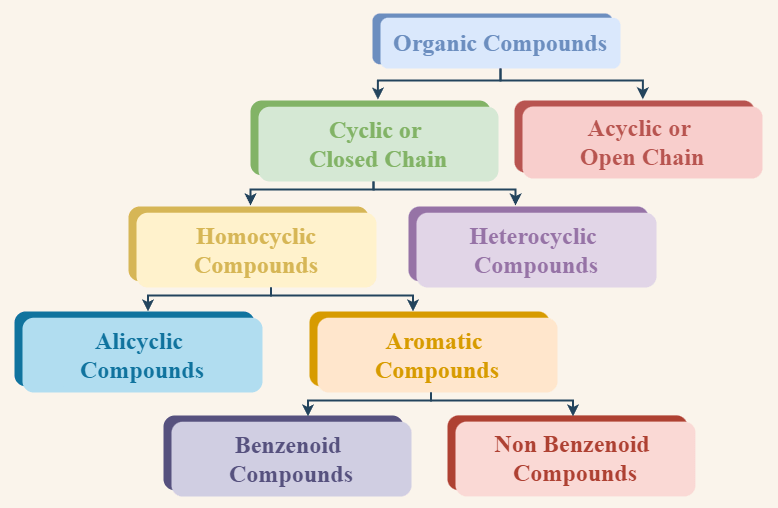
Organic compounds based on their structure are broadly categorized as:
- Open-Chain Compounds or Acyclic Compounds.
- Closed-Chain Compounds or Cyclic Compounds.
Acyclic or Open Chain Compounds
When organic compounds are formed by joining carbon atoms in the form of an open chain they are called Open Chain organic compounds. Initially, they are called Aliphatic compounds because they are derived from either animal fats or vegetable fats.
They are further classified as either straight-chain compounds or branched-chain compounds.
Straight Chain Compounds
In straight-chain compounds, the carbon skeleton is in the form of a straight chain. Examples of straight-chain include
- n-Butane: CH3−CH2−CH2−CH3
Branched Chain Compounds
In Branched chain compounds the carbon skeleton is in the form of a branched chain. Examples of branch-chain include,
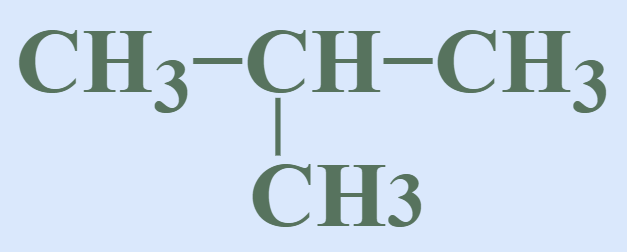
Cyclic or Closed Chain Compounds
These are the compounds in which carbon atoms are joined to form one or more rings with or without heteroatom. On the basis of heteroatoms in the cycle, they are further classified into two types
- Homocyclic Compounds
- Heterocyclic Compounds
Homocyclic or Carbocyclic Compounds
Homocyclic Compounds are cyclic compounds, in which the ring structure is formed by similar atoms of carbon. They are also called Carbocyclic compounds. No other element besides carbon exists in these compounds. Examples of Homocyclic or Carbocyclic compounds are this compound are benzene, naphthalene, tetracene, and so on.

They are further classified as,
- Alicyclic compounds: These compounds show properties similar to those of aliphatic compounds. In these homocyclic compounds, carbon atoms are linked by single bonds only. e.g. Cyclopropane, etc.
- Aromatic Compounds: These compounds contain at least one aromatic ring which resembles benzene in their chemical behaviour. e.g. Benzene, etc. Aromatic compounds are further classified into two categories which are discussed further in this article.
Heterocyclic Compounds
These compounds include one or more heteroatoms like oxygen, nitrogen, sulfur, etc. in the ring of carbon atoms. They are also further divided into two types.

- Hetero-alicyclic compounds: Alicyclic compounds which contain at least one heteroatom in the ring are called hetero-alicyclic compounds. e.g. Tetrahydrofuran (THF), etc.
- Hetero-aromatic compounds: Aromatic compounds which contain at least one heteroatom in the ring are called hetero-aromatic compounds. e.g. Furan, etc.
Hetero-aromatic compounds are classified as benzenoid compounds and non-benzenoid compounds.
Aromatic Compounds
Aromatic Compounds are a special type of organic compound that involves benzene and other ring-related compounds. They are called Aromatic compounds because these compounds carry a specific aroma, which distinguishes them from other organic compounds.
Classification of Aromatic Compounds
Aromatic compounds are highly stable organic compounds and can further be classified into two categories.
- Benzenoid Aromatic Compounds
- Non-Benzenoid Aromatic Compounds
Now, let us discuss them in detail.
Benzenoid Aromatic Compounds
Benzenoid Aromatic compounds are derivatives of benzene. These compounds are distinguished from other organic compounds as they contain one or more than one benzene ring. These compounds can be further arranged on the basis of the benzene rings fused together as
- Monocyclic
- Bicyclic
- Tricyclic
Some examples of these compounds are Phenanthrene, Naphthalene, and Anthracene.

Non-Benzenoid Aromatic Compounds
Non-benzenoid Aromatic Compounds consist of other unsaturated rings instead of benzene rings. These compounds are highly stable and some examples of these compounds are Tropolone and Azulene.

Homologous Series
A homologous series is a set of organic compounds that each include a distinct functional group and differ from one another in the molecular formula by the a-CH2-(methylene) group.
Individual series members are referred to as homologs, and they can be represented by a generic molecular formula. Homologs are ordered in increasing order of molecular mass in homologous series.
The General molecular formula is, CnH2n + 1 (Functional group)
For Example, the homologous series of alcohol is
|
Member
|
Name
|
|
CH3OH
|
Methyl alcohol
|
|
C2H5OH
|
Ethyl alcohol
|
|
C3H7OH
|
Propyl alcohol
|
|
C4H9OH
|
Butyl alcohol
|
Note: Ethyl alcohol is higher homologous of methyl alcohol but a lower homolog of propyl alcohol
Characteristics of Homologous Series
The following are the characteristics of homologous series:
- The same general formula may be used to express homologs.
- Homologs are members of the same functional group.
- Because the consecutive components’ chemical formulas change by -CH2-group, their molecular weights differ by 14 units.
- Homologs can be synthesized using comparable chemical processes.
- Homologs have chemical characteristics that are identical.
- Physical characteristics such as melting point, boiling temperature, density, solubility, and so on vary across homologs.
Classification of Organic Compounds based on Functional Group
The functional group is the part of a molecule that influences its chemical characteristics. Every organic molecule’s chemistry, regardless of size or complexity, is defined by the functional group it contains.
Organic compounds are categorized as follows based on their functional group:
Hydrocarbons
Hydrocarbons are organic compounds that consist of carbon and hydrogen atoms only. They are the simplest and most abundant organic compounds found in nature and serve as the building blocks for more complex organic molecules. Hydrocarbons are classified into two main groups that are:
- Aliphatic Hydrocarbons
- Saturated Hydrocarbons (contain only single bonds between carbon atoms)
- Unsaturated Hydrocarbons (contain one or more double or triple bonds between carbon atoms)
- Aromatic Hydrocarbons
|
Class
|
General Formula
|
Functional Group
|
Common Suffix/prefix (systematic)
|
|
Alkanes
|
R – H
|
C – C
|
-ane
|
|
Alkenes
|
R – CH = CH – R
|
C = C
|
-ene
|
|
Alkynes
|
R – C ≡ C – R
|
C ≡ C
|
-yne
|
|
Arenes
|
Ar-H
|
 |
-ene
|
Learn more about Functional Groups.
Compounds Containing Halogen
Organic halides or organohalides or simply halogenated compounds, are those organic compounds that contain halogen atoms such as fluorine, chlorine, bromine, or iodine. These compounds can be classified further:
- Alkyl Halides (R-X)
- Aryl Halides (Ar-X)
|
Class
|
General Formula
|
Functional Group
|
Common Suffix/prefix (systematic)
|
|
Alkyl Halides
|
R – X
|
– X
|
-halide
|
|
Aryl Halides
|
ArX
|
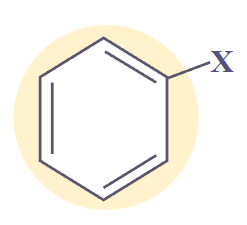 |
halo-
|
Learn more about Halogen.
Compounds Containing Oxygen
As carbon and hydrogen are the most abundant elements in the
- Alcohol (R-OH)
- Phenol (Ar-OH)
- Aldehyde (R-CHO)
- Ether (R-O-R)
- Carboxylic Acid (R-C(=O)-OH)
|
Class
|
General Formula
|
Functional Group
|
Common Suffix/prefix (systematic)
|
|
Alcohols
|
R – OH
|
– OH (hydroxyl)
|
-ol
|
|
Phenols
|
Ar – OH
|
– OH (Phenolic)
|
-ol
|
|
Ethers
|
R – O – R’
|
– C – O – C –
|
ether
|
|
Aldehydes
|
RCHO
|
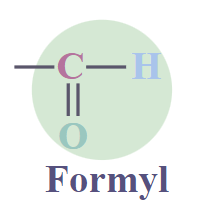 |
-al
|
|
Carboxylic acid
|
RCOOH
|
 |
-ic acid
|
Carboxylic Acid Derivatives
Carboxylic acid derivatives are a class of organic compounds that are structurally related to carboxylic acids. They contain a carbonyl group (C=O) bonded to a heteroatom, such as oxygen, nitrogen, or sulfur.
- Ketone (R-C(=O)-R)
- Ester (R-COOR’).
- Amides (R-CO-NR’R”)
- Acid Halides (R-CO-X)
- Anhydride (R-C(=O)-O-C(=O)-R’)
|
Class
|
General Formula
|
Functional Group
|
Common Suffix/prefix (systematic)
|
|
Ketones
|
RR’C = O
|
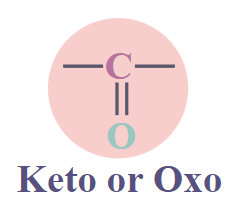 |
-one
|
|
Esters
|
RCO2R’
|
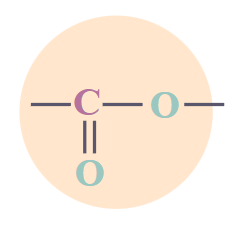 |
-ate
|
|
Amides
|
RCONHR’
|
 |
-amides
|
| Acid Halides |
RCOX |
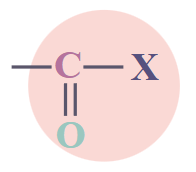 |
Alkyl Halide
|
| Anhydride |
RCOOCOR’ |
 |
-oic anhydride
|
Compounds Containing Nitrogen
Nitrogen is the seventh most abundant element in the universe and is present in a wide range of compounds, including amino acids, DNA, and proteins thus compounds containing nitrogen are a diverse and essential class of organic compounds. Some compounds containing nitrogen are:
- Amines (R-NH2)
- Cyanide or Nitrile (R-C≡N)
- Nitro Compounds (R-NO2)
- Imines (R-C=N-R’)
Learn more about Nitrogen.
|
Class
|
General Formula
|
Functional Group
|
Common Suffix/prefix (systematic)
|
|
Amines
|
RNH2 (Primary)
RNHR’ (Secondary)
RNR’R” (Tertiary)
|
-NH2
-NH-
-N=
|
-amine
|
|
Nitriles
|
RC ≡ N
|
– C ≡ N
|
-nitrile
|
|
Nitro Compounds
|
ArNO2
|
 |
nitro-
|
|
Imines
|
R2CNR’
|
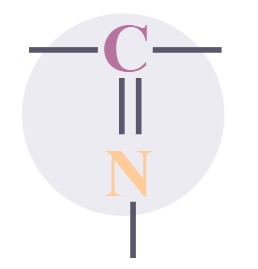 |
-imine
|
Compounds Containing Sulfur
Sulfur-containing compounds are molecules that contain one or more sulfur atoms in their chemical structure. Sulfur is a non-metallic element with the atomic number 16 and is found in many minerals and organic materials. Some organic compounds containing Sulfur are:
- Thiols (-SH)
- Sulfides or Thioethers (R-S-R’)
- Sulfoxides (R-S(=O)-R’)
- Sulfones (R-S(=O)2-R’)
- Thiocyanates (R-S-C≡N)
|
Class
|
General Formula
|
Functional Group
|
Common Suffix/prefix (systematic)
|
|
Thiols
|
RSH
|
-SH
|
mercapto-
|
|
Sulfides or Thioethers
|
R-S-R’
|
– S –
|
-sulfide
or
alkylthio-
|
|
Sulfoxides
|
RSOR’
|
 |
-sulfoxides
or
-sulfinyl-
|
|
Sulfones
|
RSOOR’
|
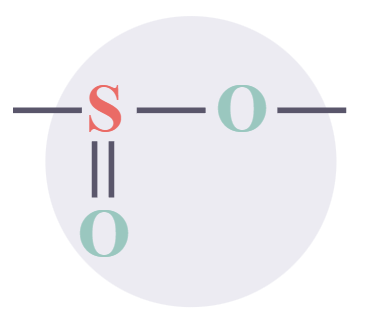 |
-sulfonyl-
|
|
Thiocyanates
|
RSCN
|
 |
-thiocyanate
|
Note: In above table R – Represents Alkyl group and Ar – represents Aryl group.
Read More,
Sample Questions on Organic Compounds
Question 1: Write a note on the homologous series.
Answer:
Homologous series is a series of organic compounds containing characteristics functional group and the successive member differ from each other by a -CH2 group.
Organic compounds form homologous series such as alkanes, akenes, alknes, alcohol, amines, etc.
Question 2: What are volatile organic compounds?
Answer:
Volatile organic compounds are specific organic compounds with high vapour pressure and low water solubility. Volatile Organic Compounds are human-made chemicals that are used in the manufacturing of paints, pharmaceuticals, and refrigerants.
Question 3: Write the structural formula of the first 4 members of the homologous series of the following compounds
- Aldehydes
- Carboxylic acid
Answer:
Aldehydes
- H -CHO (methanal)
- CH3 – CHO (ethanal)
- CH3 – CH2 – CHO (propanal)
- CH3 – CH2 – CH2 – CHO (butunal)
Carboxylic Acid
- H – COOH (methanoic acid)
- CH3 – COOH (ethanoic acid)
- CH3 – CH2 – COOH (propanoic acid)
- CH3 – CH2 – CH2 – COOH (butanoic acid)
Question 4: Why are most organic compounds insoluble in water?
Answer:
Most organic compounds are insoluble in water because they are non-polar molecules and water is a polar solvent.
Question 5: What organic compounds are used in medicine?
Answer:
Organic Compounds used in medicinal products are atorvastatin, fluticasone, and clopidogrel other than that proteins, fats and carbohydrates are also sometimes used as medicines.
Question 6: How to identify an organic compound?
Answer:
Organic compounds can easily be identified by carbon atoms as all organic compounds contain carbon atoms.
Organic Compounds – FAQs
Define Organic Compounds.
Organic compounds are compounds which contain Carbon atoms linked with other atoms through covalent bonds. They are called organic compounds because initially they are derived from organic materials.
What are Some Examples of Organic Compounds?
Some common examples of organic compounds include carbohydrates (such as glucose and fructose), proteins (such as enzymes and hemoglobin), lipids (such as fats and oils), and nucleic acids (such as DNA and RNA).
Which is the Essential Element in all Organic Compounds?
Carbon is an essential element in all organic compounds. Other than carbon hydrogen and oxygen are most abundent in the organic compounds.
How are Organic Compounds Classified?
Organic Compounds can be classfied as follows:
- Aliphatic hydrocarbons
- Saturated hydrocarbons
- Unsaturated hydrocarbons
- Aromatic hydrocarbons
- Compounds Containing Halogen
- Alkyl Halides (R-X)
- Aryl Halides (Ar-X)
- Compounds Containing Oxygen
- Alcohol (R-OH)
- Phenol (Ar-OH)
- Aldehyde (R-CHO)
- Ether (R-O-R)
- Carboxylic Acid (R-C(=O)-OH)
- Carboxylic Acid Derivatives
- Ketone (R-C(=O)-R)
- Ester (R-COOR’).
- Amides (R-CO-NR’R”)
- Acid Halides (R-CO-X)
- Anhydride (R-C(=O)-O-C(=O)-R’)
- Compounds Containing Nitrogen
- Amines (R-NH2)
- Cyanide or Nitrile (R-C≡N)
- Nitro Compounds (R-NO2)
- Imines (R-C=N-R’)
- Compounds Containing Sulfur
- Thiols (-SH)
- Sulfides or Thioethers (R-S-R’)
- Sulfoxides (R-S(=O)-R’)
- Sulfones (R-S(=O)2-R’)
- Thiocyanates (R-S-C≡N)
What is the Difference Between Organic and Inorganic Compounds?
Organic compounds are molecules composed of carbon atoms bonded to hydrogen and other elements, while inorganic compounds do not contain carbon-hydrogen bonds. Some common examples of inorganic compounds include water, salt, and minerals.
Are all Organic Compounds Safe?
No, not all organic compounds are safe. Some organic compounds can be toxic, carcinogenic, or otherwise harmful to human health and the environment. It is important to handle organic compounds with care and to follow appropriate safety protocols when working with them.
What are the Uses of Organic Compounds?
Organic Compounds are used in a variety of industries such as foods and packaging, biofuel, fossil fuels, drugs-medicine and others.
Like Article
Suggest improvement
Share your thoughts in the comments
Please Login to comment...