Classification of Matter
Last Updated :
30 Jun, 2021
In our surroundings, we come across things that are different in shape, size, height, structure, and texture. According to scientists everything in this universe is made up of a material called ‘matter’. We can see that matter occupies some space and mass, in another way we can say that matter has some ‘volume’ and ‘mass’. However, in ancient times there was no clue to the scientists therefore at that time Indian Philosophers believed that everything in this universe either living or non-living can be divided into ‘panch tattvas i.e. Air, Water, Fire, Sky, and Earth.
Now in this article, we will discuss about matter, its physical properties on the basis of its classification and will also study its types i.e. solid, liquid, and gas in brief.
What is Matter?
Matter refers to the material things around us which have mass, occupied space and its presence can be felt by any one or more of our five senses. Everything in this universe is made up of material that scientists have named ‘matter’.
For example- the water, air, stars, plants, a small drop of water, or even a small sand particle is matter, but emotions like love, hate, hurt, etc. are feelings but not matter.
The matter is anything that occupies space and has mass.
Space: Space occupied by a substance is known as ‘volume’.
Mass: The quantity of metal present in an object is called its ‘mass’.
Classification of Matter
Early Indian philosophers classified matter in the form of 5 basic elements as Air, Earth, Fire, Sky, and Water called ‘panch tatva’. Ancient Greek philosophers also applied a similar classification of matter. Classification on the basis of physical properties and chemical properties. What is observed and measured without changing the chemical identity of samples like color, length and volume are physical properties, and what is observed and measured that simply change its chemical identity are called chemical properties.
On the basis of the physical state of matter, it is classified as solid, liquid, and gas. On the basis of chemical composition, the matter is classified as pure substance and mixture. Pure substances may be elements and compounds whereas Mixtures may be homogeneous mixture and heterogeneous mixture.
Physical Nature of Matter
- A matter is made up of very tiny particles which may be atoms or molecules, for example- when we dissolve a spoon of salt or sugar in a glass of water.
- Matter is made up of particles– The particles of matter are very small beyond imagination these particles have space between them.
- Particles of matter are in continuous motion– This is because of the kinetic energy possessed by the particles which increases on increasing the temperature and so particles move much faster.
- Particles of matter attract each other by attractive forces– The attractive forces bind the particles of matter in a single body and also lead to the arrangement of particles. The particles of matter have a force acting between them.
Note: Both sugar and salt are made up of very small particle which break off from each sugar granule and get disappeared in water on dissolving, that’s why solution tastes sweet, but particle cannot be seen, this shows that water have enough space to adjust (dissolve) tiny sugar or salt particle.
Characteristics of Particles of Matter
- Particles of matter have space between them.
- Particles of matter are continuously moving.
- They possess some energy called Kinetic energy as the temperature rises the kinetic energy of the particles increases and hence particles move fast.
- Particles of matter attract each other, a force of attraction exists between the particles known as an intermolecular force of attraction.
- The force is called Cohesive Force.
States of Matter
- Solid- Solid has a definite shape and definite volume. For example- ice cube and wood.
- Liquid- A liquid has a definite volume but no definite shape. For example- water and milk
- Gas- A gas has neither a definite shape nor a definite volume. For example- Nitrogen, Oxygen, etc.
- Plasma– Plasma is considered the fourth state of matter. Plasma is a mixture of free electrons and ions. It occurs naturally in the stars. Inside the stars, the temperature is so high that the atoms break down. And, this mixture of free electrons and ions in a state is called plasma. The sun and the other Stars glow because of the pressure of Plasma in them. Plasma can also be made on the earth by passing electricity through gases at less temperature plasma make the fluorescent tube glow.
- BE condensate(bose-einstein)– In 1921 Indian scientist Satyendra Nath Bose did some calculations for the fifth state of matter. On the basis of their calculation, Albert Einstein predicted the existence of a new state of matter. Fifth state of matter was achieved finally by three scientists Cornell, Kellerie, and Wieman of USA cooling gas in extremely low density to super low temperature.
Now, let’s study the properties of the three major states of the matter in detail
Properties of Solid
- Definite shape, size, volume, and distinct boundaries.
- Negligible compressibility.
- Solids have a tendency to maintain their shape when subjected to outside force.
- They are rigid, difficult to change their shape.
- Mass per unit volume of a substance is called density.
- Intermolecular force is high in solids.
- Kinetic energy is very low in solids.
- Solids do not possess the property of diffusion.
- A rubber band is a solid because it can change its shape under force and regain its shape when force is removed if excessive force is applied it breaks. This is exceptional case of the solid.
- Salt and sugar take the shape of the container in which they are placed, but the shape of their crystals does not change, so they are solids.
Properties of Liquid
- Do not have a definite shape or distinct boundaries but have a fixed volume.
- They can be compressed.
- In Science the common name of gases and liquids is fluid.
- Liquids are not Rigid but have the property of flowing that’s why liquids are called fluids.
- Intermolecular force is less than the solids.
- In liquid kinetic energy is more than solid.
- Liquid has moderate density.
- Liquids possess the property of diffusion.
- They can take any shape.
- The gases Oxygen and Carbon dioxide from the atmosphere diffuse and dissolve in water. Due to these gases, aquatic plants and animals are able to survive.
- Diffusion is much more in liquid than in solid due to the free movement of particles of liquid.
Properties of Gas
- Gas is neither a definite shape nor a definite volume.
- They can be compressed much.
- Gases have maximum fluidity and less rigidity.
- The intermolecular force of attraction is least.
- The kinetic energy of its particles is maximum.
- Gases have no density.
- They can take any shape.
- Very fast diffusion.
- The particles in a gas are free to move and hence gases can flow in any direction. They occupy all the volume available to them.
- LPG = Liquefied Petroleum Gas and CNG = Compressed Natural Gas that is fuel in vehicles.
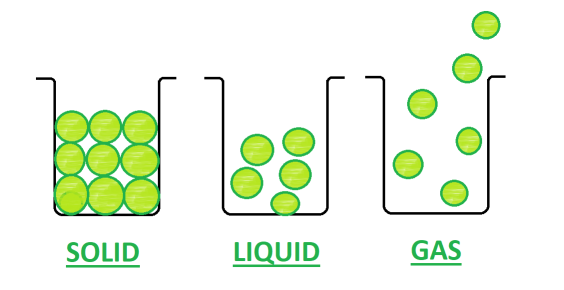
Difference Between Solid, Liquid and Gas
| |
SOLID |
LIQUID |
GAS |
| 1. |
Solids have a fixed volume and a definite shape. It has distinct boundaries. |
Liquids have a fixed volume but have no fixed shape. They take the shape of containers but do not fill it. |
Gases have neither a fixed shape nor a fixed volume. They take the shape and volume of the vessel. They fill the container. |
| 2. |
Solids are rigid and are not compressible. |
Like solids, liquids cannot be compressed much. |
Gases are easily compressible. |
| 3. |
Space and movement of a particle is minimum. |
Movement of particles is intermediate. |
Movement of particles is maximum. |
| 4. |
Solids do not flow and do not diffuse. |
Liquids generally flow easily and can diffuse more than solids. |
Gases flow easily and have a strong tendency for diffusion. |
| 5. |
Solid have high densities. |
They are generally less dense than solids. |
Gases have very low densities and are very light. |
| 6. |
Solids stay where they are placed. |
Liquids can be poured. |
Gases spread out quickly. |
| 7. |
Interparticle force of attraction is strongest in solids. |
Interparticle forces are weaker. |
Interparticle force of attraction is weakest. |
| 8. |
Solid posses rigidity. |
Liquid posses fluidity. |
Gases posses the highest fluidity. |
| 9. |
For example- ice, wood, sugar, etc. |
Ex- water, juice, etc. |
Ex- Oxygen, Nitrogen, etc. |
Sample Questions
Question 1: What is diffusion?
Solution:
Intermixing of particles of two different type of matter on their own is called diffusion.
OR
The spontaneous intermixing of particles of two different types matter is called diffusion. The rate of diffusion becomes faster with increase in temperature because at high temperature the particles have more energy and hence move faster.
For example -aroma of perfume, smell of hot sizzling food
Question 2: What is inter-particle force of attraction?
Solution:
Matter have interparticle force of attraction. This force keeps the particles together. It depends on nature of matter.
Question 3: How small are these particles the particles of matter?
Solution:
The particles of matter are so small that we can’t even see them with the naked eyes.
Question 4: What happens when a swollen balloon is poked with a pin?
Solution:
It will burst and diffusion will take place.
Question 5: What is the reason behind the observation, that liquid can not be compressed while gasses can?
Solution:
This is because of the phenomenon of the intermolecular space which is very large in gasses that’s why have capacity to compress while liquids have less intermolecular space therefore can’t be compressed.
Question 6: Why solids have a fixed shape?
Solution:
Solids have fixed shape because their intermolecular force is very strong that’s why always holds up a regular geometrical structure.
Question 7: Arrange the following substances in decreasing order of the intermolecular force of attraction.
- Carbon-dioxide
- Sugar
- Juice
Solution:
Sugar > Juice > Carbon-dioxide
Question 8: What is the Brownian Movement?
Solution:
The zigzag movement of the small particles suspended in a liquid or gas is called Brownian movement.
Like Article
Suggest improvement
Share your thoughts in the comments
Please Login to comment...