Change of State of Matter
Last Updated :
01 Apr, 2024
When cubes of ice melt into water or liquid boils into vapor, you may have seen changes in states of matter, but have you ever wondered why the substances change their form? When matter loses or gains energy, it changes its condition. When a substance gains energy, its molecules or atoms move faster, and extra kinetic energy pushes the particles at a sufficient distance apart from one another that their shape changes.
Typically, this energy is called thermal energy or heat. Let us look at the science supporting the shifting states of matter in this post.
Change of state
A physical change in a matter is referred to as a change of condition. They are reversible alterations that do not entail any modifications to the matter’s chemical composition. Deposition, melting, sublimation, freezing, vaporization and condensation are examples of state transitions. The modifications are depicted in the diagram below.
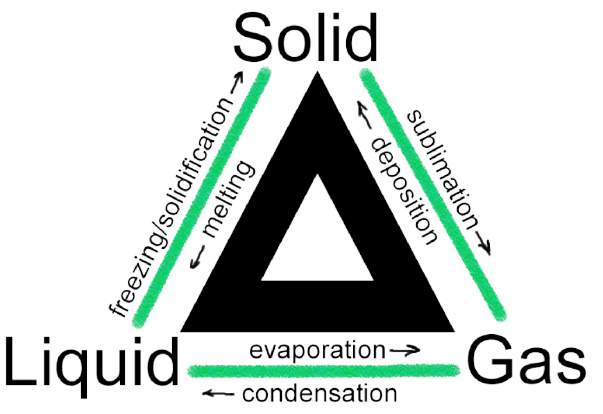
Must Check
What Causes Phase Changes?
Change of phase occurs when a system’s pressure or temperature changes. The contact between particles increases as pressure or temperature rises. Similarly, when the temperature falls, atoms and molecules find it simpler to form a more hard structure.
1. Change of phase between solids and liquids
In a tray, how would you form ice cubes? To begin, the tray is filled with water from a faucet. The tray would then be placed in the refrigerator’s freezer compartment. The freezer is really frigid. So, what’s next?
- Freezing: Heat is transferred from the warmer tray to the freezer’s colder air. The warm water in the freezer loses heat to the chilly air. This heat transfer continues until the particles lose all their energy to move past each other. They are compelled to remain in fixed places because of the force of attraction between them. Water (liquid) is transformed into ice (solid) in this manner. Therefore, freezing is the process of the transformation of water into solid. The freezing point is the temperature at which the transformation happens.
- Melting: The ice cubes would absorb energy from the warmer air surrounding them if they were removed from the freezer and placed in a warm area. The absorbed energy would help them to deal with the force of attraction that bound them together, allowing them to break free from their ice-like grip. Melting is the transformation of a solid into a liquid state. Melting point is the temperature at which a solid transforms into liquid.
2. Change of phase between gases and liquids
The water warms up when you fill a pot with cold tap water and heat it on a hot cooktop. Heat energy flows from the cooktop to the pot, where it is absorbed by the water. What will happen to the water after that?
- Vaporization: If the water is sufficiently heated, it will begin to boil. In the boiling water, water vapor bubbles develop. This occurs when liquid water particles obtain enough energy to overcome the force of attraction between them and transition to a gaseous form. The bubbles rise through the water and exit as steam, which escapes from the pot. Vaporization is the process through which a liquid boils and transforms into a gas. The boiling point of a liquid is the temperature at which it boils.
- Condensation: The mirror is prone to fog up when you take a hot shower in a closed bathroom. You might be wondering why this occurs. As hot water from the shower evaporates, it cools and loses energy when it comes into touch with colder surfaces, such as the mirror. The energy required by the colder water particles to overcome the forces of attraction between them is no longer available. They clump together and create liquid water droplets. Condensation is the process through which gas transforms into a liquid.
3. Change of phase between gases and solids
Solids that transform to gas must first pass through a liquid condition. Solids may, on the other hand, transform from solid to gas without passing through the liquid state. The opposite change can also happen. Gases can sometimes convert straight to solids.
- Sublimation: Sublimation is the process of solids converting straight to gases. When solids absorb enough energy, the forces of attraction between them are totally eliminated. Sublimation occurs when solids, such as dry ice, are heated. We can’t forget about air fresheners while we’re talking about sublimation examples. Solid air fresheners (such as those used in toilets) have a distinction for being exquisite in nature.
- Deposition: Deposition is a phase change in which a gas becomes a solid without first passing through the liquid phase. Thermodynamics governs the process of deposition. Sublimation is the inverse of deposition, hence deposition is also referred to as desublimation. For example, water vapour in the air transforms to small ice crystals when warm moist air inside a house comes into touch with a freezing cold windowpane.
Conclusion
- Every item in existence experiences a state transition, which you should be aware of. It’s simply a matter of how much heat is applied to the material.
- Everything on our planet can be manipulated to change its condition if enough heat is provided. The point is, not all substances must take the solid-liquid-gas route.
- Some compounds can spontaneously transit from a solid to a gaseous state without passing through a liquid stage.
- Sublimation is the term for this process. The dry ice, element iodine, and high-quality coal, which burns and sublimates into vapor at high temperatures, are all examples of sublimation.
- In total, we have witnessed six changes in states of matter so far.
Sample Problems
What does it take to alter the state of matter?
It specifies a matter’s changing condition when you add or remove heat from it. When you apply heat to a substance, it begins to melt. When the heat is removed, though, it begins to solidify. The movement of particles causes a change in the state when heat is added or removed from the substance. The atoms in a substance are always moving at various speeds. They gain speed as a result of the heat.
Explain the sentence “The liquid state is a transitional condition between the gaseous and solid states.”
A liquid has a lower force of attraction than solids but a higher force than a gas. A liquid has more inter-particle gaps than solids and less inter-particle spaces than a gas. This demonstrates that liquids are a transitional state between solids and gases.
What are the two types of changes that can happen in a situation?
Physical and chemical alterations are the most common changes that matter endures. There is no change in the identity of a matter after physical transformation. On the other hand, its shape, size, and status vary. However, if a chemical change occurs, it cannot be reversed or is only reversible under particular conditions.
What is the mechanism through which gases become solids directly?
Deposition is the process through which a gaseous state transforms into a solid state without transitioning to a liquid state.
How many different states of matter are there?
Matter is anything in our surroundings that has its own mass and occupies a certain amount of space. It can also be defined as objects that we can taste, touch, or smell. For instance, a chair, air, water, gold, and so on. An atom is the smallest particle of matter.
There are five forms of matter known to us out of which three are found under normal conditions. These are:
- Solid
- Liquid
- Gas
- Plasma
- Bose Einstein
Solid, liquid and gas are common forms out of these matters.
Like Article
Suggest improvement
Share your thoughts in the comments
Please Login to comment...