Catalysis in Chemistry is defined as the process in which the rate of the reaction is influenced by the presence of some specific substance. These specific substances are called Catalysts. The catalyst is never consumed during the chemical reaction.
A catalyst changes the activation energy of the reaction, i.e. the energy required for occurring the reaction. and thus alters its rate. In general, a catalyst lowers the activation energy of the chemical reaction and this process is called the Catalysis.
What is Catalysis in Chemistry?
Catalysis is a process that can speed up and slow down a chemical reaction. The substance which cause the change in rate of recation without being consumed at the end of the reaction.
Catalysis in chemistry is a process that increase or decrease the rate of a chemical reaction caused by the inclusion of a catalyst, which is not consumed in the chemical reaction and is used multiple times.Every chemical reaction proceeds after overtaking a certain energy and this energy is called the Activation Energy of the reaction. The catalyst alters the Activation Energy of the chemical reaction and increases and decreases the rate of the reaction.
Catalysis Definition
Catalysis is the generally defined as the process that increases the rate of the reaction and makes the chemical reaction faster.
Catalysis Example
The example of Catalysis is mentioned in the following reactions:
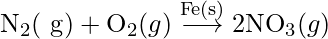
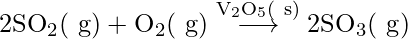
Mechanism of Catalysis
We know that a catalyst decreases the activation energy of the reaction and increases the rate of the reaction. The mechanism of the catalysis is defined by a five-step process that is added below,
Step 1: Firstly reactant molecule is diffused on the catalytic surface.
Step 2: Now the reactant molecules get adsorped on the catalytic surface.
Step 3: Then the intermediate molecules are formed on the catalytic surface.
Step 4: Then desorption of the product molecule do formed by the catalytic surface.
Step 5: Now at last the diffusion of product molecules from the catalytic surface to form Final product.
Types of Catalysis
Catalysis are easily categerozied into two different categories that are,
- Homogeneous Catalysis
- Hetrogenous Catalysis
Homogeneous Catalysis
When the reactants and catalyst are in the same phase with respect to each other (i.e. solid, liquid or gas). It is claimed that the catalysis is homogeneous.
Some examples of homogeneous catalysis are listed below.
- In the lead chamber method, sulphur dioxide is oxidized to sulphur trioxide with oxygen in the presence of nitrogen oxides as a catalyst.
2SO2(g) + O2(g) → 2SO3(g) {In presence of NO(g)}
The reactants, products, and catalysts are all in the same phase or gaseous state.
- Sugar hydrolysis is catalyzed by the H+ ions produced by sulphuric acid.
C12H22O11(l) + H2O(l) → C6H12O6(l) + C2H12O6(l) {In presence of H2SO4(l)}
Heterogeneous Catalysis
Heterogeneous catalysis is a catalytic process in which the reactants and catalysts are in different phases.
The following are some examples of heterogeneous catalysis-
- In the contact method for the synthesis of sulphuric acid, oxidation of sulphur dioxide into sulphur trioxide with platinum metal or vanadium pentaoxide as a catalyst. While the reactants are gaseous, the catalyst is solid.
SO2(g) + O2(g) → 2SO3(g) {In presence of Pt(s)}
- In Haber’s process, nitrogen and hydrogen are combined to produce ammonia in the presence of finely split iron.
N2(g) + 3H2(g) → 2NH3(g) {In presence of Fe(s)}
- In Ostwald’s procedure, ammonia is converted to nitric oxide in the presence of platinum gauze as a catalyst.
4NH3(g) + 5O2(g) → 4NO(g) + 6H2(g) {In presence of Pt(s)}
Apart form this there are also two types of catalysis that are,
- Positive Catalysis
- Negative Catalysis
Positive Catalysis
When a foreign substance accelerates the pace of a reaction, it is referred to as a positive catalyst, and the phenomenon is known as positive catalysts.
The following are some examples of Positive catalysis,
- Decomposition of H2O2 in presence of colloidal platinum.
2H2O2(l) → 2H2O(l) + O2(g) {In presence of Pt}
Negative Catalysis
Certain substances, when added to the reaction mixture, slow down rather than speed up the reaction rate. Negative catalysts or inhibitors are used in this process, which is known as negative catalysis.
The following are some examples of Negative catalysis
- If some alcohol is added to chloroform, the oxidation by air is slowed.
2CHCl3(l) +O2(g) → 2COCl2(g) + 2HCl(g) {In presence of Alcohol(l)}
Auto-Catalysis
One of the products functions as a catalyst in some processes. The reaction is slow in the beginning, but after the products are formed, the rate of reaction accelerates. Auto-catalysis is the term for this type of event.
Some examples of homogeneous catalysis are listed below.
- When nitric acid is poured on copper, the reaction is slow at first, but it quickly accelerates due to the creation of nitrous acid, which works as an auto-catalyst during the process.
Acid-Base Catalysis
H+ or OH– ions, according to Arrhenius and Ostwald, operate as catalysts.
- Conversion of acetone into diacetone alcohol,
CH3COCH3(l) + CH3COCH3(l) → CH3COCH2.C(CH3)2OH(l) (In presence of OH–)
- Decomposition of Nitramide,
NH2NO2(l) → N2O(g) + H2O(l) (In presence of OH–)
Poison Catalysis
Poison catalysis is the process in which we add catalyst to stop the chemical reaction. Thus, catalyst poison is defined as the substance that decreases the effectiveness of the catalyst added and stops the chemical reaction.
In the Habber’s Process of manufacturing of Ammonia, Fe is used as catalyst, and oxygen containing compounds such as Carbon Monooxide acts as poison to the catalyst.
N2 + 3H2 (Fe/CO)→ 2NH3
Promoters are the chemical substance that increases the effectiveness of the catalyst in the chemical reaction. Promoters individually have very little or no effect on the chemical rection.
N2 + 3H2 (Fe/Mo)→ 2NH3
In the above reaction, Fe is the catalyst and Mo is the promoter that increases the rate of the reaction.
Characteristics of Catalysis
Various properties/characterstics of the catalyst are added below,
- At the end of the reaction, the mass and chemical makeup of a catalyst remains unchanged.
- The reaction cannot be started by the catalyst.
- In most cases, the catalyst is quite specific.
- The catalyst has no ability to alter the state of equilibrium.
- Changes in temperature affect the pace of catalytic reactions, just as they do in the absence of a catalyst.
Features of Solid Catalysis
Various features of the solid catalysis of any chemical reaction are,
Catalyst Activity
Catalyst activity refers to how quickly they may speed up chemical reactions. In some reactions, the rate of acceleration can reach 1010 times. In the presence of platinum as a catalyst, the reaction between H2 and O2 to generate H2O, for example, occurs with explosive violence. H2 and O2 can be held indefinitely without undergoing any reactions in the absence of a catalyst.
Catalyst Selectivity
It is possible for catalysts to direct reactions to produce certain products (excluding others).
Theories of Catalysis
Various theories are present that explain the working of the catalyst and some of them are,
- Collision Theory of Homogeneous Catalysis
- Adsorption Theory of Heterogeneous Catalysis
Collision Theory of Homogeneous Catalysis
According to the Collision Theory, A reaction happens as a result of effective collisions between the reacting molecules The molecules must have a minimum level of energy known as activation energy (Ea) in order to collide effectively. Following the collision, the molecules form an activated complex, which then dissociates to reveal the product molecules. The catalyst creates a novel pathway that requires less activation energy.
Adsorption Theory of Heterogeneous Catalysis
Adsorption of reactants on the surface of the catalyst is usually how heterogeneous catalytic reactions work. The diffusion theory of catalysis can be used to describe the mechanism of such surface reactions.
Zeolites (Shape-Selective Catalysis)
Zeolites are aluminosilicates of the general formula, Mn[AlO2]x.(SiO2)y.mH2O, where, M may be a simple cation like Na, K or Ca, n is the charge on the simple cation, m is the number of molecules of water of crystallization.
They have a β cage, which looks like a honeycomb. The shape and size of the reactant and product molecules influence the processes catalyzed by zeolites. This is why these reactions are referred to as shape-selective catalysis.

In the petrochemical industry, zeolites are commonly utilized as catalysts for the cracking and isomerization of hydrocarbons. The ZSM-5 zeolite catalyst is a widely used zeolite catalyst in the petroleum sector (zeolite sieve of molecular porosity 5). It transforms alcohols straight to gasoline by dehydrating them first, resulting in a mixture of hydrocarbons. In the softening of hard water, hydrated zeolites are used as ‘ion-exchangers.’
Read More,
Catalysis: FAQs
1. What is Catalysis Class 12?
“A catalyst is a substance that can speed up and slow down a chemical reaction without itself being consumed in the chemical process and this phenomenon is called catalysis.”
2. What is Catalysis Example?
Example of Catalysis, Potassium Permagnet (KMNO4) acts as catalyst in the decomposition of Hydrogen Peroxide.
2H2O2 → 2H2O + O
3. What are the 3 Types of Catalysis?
The 3 basic types of catalysis are,
- Hetrogenous Catalysis
- Homogenous Catalysis
- Enzymatic Catalysis
4. What is the Principle of Catalysis?
The basic principle of the catalysis is that any catalyst alters the activation energy of the chemical reaction altering the rate of the chemical reaction.
5. What are the Properties of Catalysts?
Various important properties of catalysts are,
- Catalyst never initiate a chemical reaction.
- Catalyst never get consumed in the chemical reaction.
- Catalyst does not change the equilibrium state of the reaction.
6. What are the Uses of Catalysis?
Catalyst are used to lower the activation energy of various chemical reaction and helps us increase the rate of some specific reactions.
Like Article
Suggest improvement
Share your thoughts in the comments
Please Login to comment...