Alkynes – Definition, Structure, Preparation, Properties
Last Updated :
17 Nov, 2022
A hydrocarbon is an organic molecule made completely of hydrogen and carbon in organic chemistry. Hydrocarbons are an example of hydrides in group 14. Hydrocarbons are colourless, hydrophobic, and have just a faint odour. It’s impossible to generalise further due to their varied molecular architectures. The burning of fossil fuels, including fuel production and combustion, accounts for the majority of anthropogenic hydrocarbon emissions. The emissions of plants are a natural supply of hydrocarbons such as ethylene, isoprene, and monoterpenes.
Alkynes
In natural science, alkynes are the unsaturated hydrocarbons having something like a triple connection between carbon-carbon particles (– C≡C–). The overall recipe of this unsaturated hydrocarbon is CnH2n–2, where n–2, 3, 4, 5… .
Alkynes are customarily known as acetyls. Like different hydrocarbons, alkynes are additionally hydrophobic.
Hydrocarbons are the simplest organic compounds, with simply carbon and hydrogen as constituents. These can be found in nature in the shape of objects that we use every day. As a result, hydrocarbons are thought of as the organic compounds’ parents. All other compounds, on the other hand, are thought to have been made by substituting functional groups for one or more hydrogen atoms.
Structure of Alkynes
A carbon-carbon triple bond comprises of one in number σ bond and two feeble π bonds.

Structure of Alkynes
Nomenclature of Alkynes
The overall recipe of this unsaturated hydrocarbon is CnH2n–2, where n–2,3,4,5… … . As per the IUPAC framework, the names of alkenes have a postfix-yne, and the prefix relies upon the number of carbon particles. A few names of the alkynes are as per the following:
| Number of carbon atoms |
Formula |
IUPAC name |
|
2
|
C2H2
|
Ethyne
|
|
3
|
C3H4
|
Propyne
|
|
4
|
C4H6
|
Butyne
|
|
5
|
C5H8
|
Pentyne
|
|
6
|
C6H10
|
Hexyne
|
|
7
|
C7H12
|
Heptyne
|
|
8
|
C8H14
|
Octyne
|
|
9
|
C9H16
|
Nonyne
|
|
10
|
C10H18
|
Decyne
|
Isomerism in Alkynes
The structural isomerism of alkynes is as follows:
- Chain Isomerism: Chain isomerism occurs in alkynes with five or more carbon atoms owing to various carbon chain configurations. As an example,

Chain Isomerism
- Position isomerism: In this case, the isomers differ in terms of the triple bond’s location. The two isomers of butene, for example,

Position Isomerism
- Functional Isomerism: Dienes have two double bonds, while alkynes are functional isomers of dienes.

Functional Isomerism
- Ring Chain Isomerism: With cycloalkanes, alkynes display ring chain isomerism. As an example,

Ring Chain Isomerism
Methods of Preparation of Alkynes
The following are some of the most common alkyne preparation methods:
- By Dehydrogenation of Dihaloalkanes: Dehydrohalogenation of vicinal-di haloalkanes in an alcoholic solution of potassium hydroxide can be used to make alkynes. As an example,
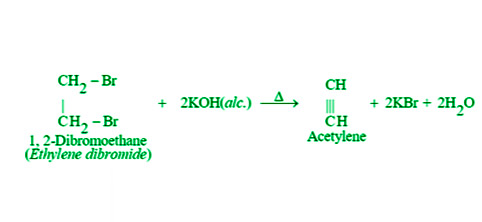
- By Dehalogenation of Tetrahalides: When tetra haloalkanes are heated in methanol with zinc dust, they dehalogenate and produce alkynes. As an example,
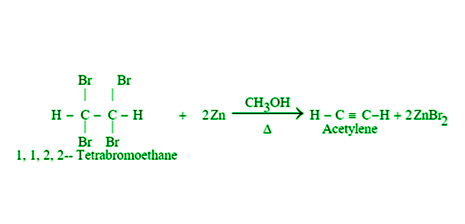
- By Dehalogenation of Haloforms: When chloroform and iodoform are heated with silver powder, they dehalogenate and yield ethyne.
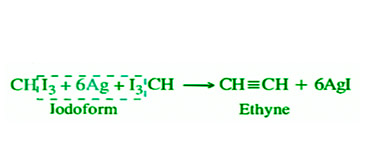
- Synthesis from Carbon and Hydrogen: Steam of hydrogen gas may similarly be used to make ethyne by passing it through an electric arc formed between two carbon electrodes. Berthelot synthesis is the name for this method.
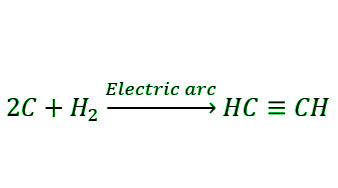
Physical Properties of Alkynes
The properties of alkynes essentially follow the similar examples to those of alkanes and alkenes.
- Alkynes are unsaturated carbon that shares triple security at the carbon site
- All alkynes are scentless and dismal except for ethylene which has a slight particular smell.
- The initial three alkynes are gases, and the following eight are fluids. All alkynes higher than these eleven are solids
- Alkynes are somewhat polar in nature
- The edge of boiling over and liquefying point of alkynes increments as their atomic design becomes greater. The edge of boiling over increments with expansion in their sub-atomic mass
- Additionally, the limits of alkynes are marginally higher than those of their relating alkenes, because of an additional one bond at the carbon site.
Chemical Properties of Alkynes
Acidic nature
Going to the synthetic properties of alkynes, we start with their marginally acidic nature. Presently Alkynes are marginally electronegative in nature. The triply fortified carbon particles in alkynes are sp hybridized, Whereas like in alkanes the single bond iotas are sp3 hybridized, causing the distinction in the electronegativity. This makes it simpler for them to draw in the common electron pair of the C-H bond. So when we respond to a solid base like NaNH2 with ethyne, we will get sodium acetylide and freed hydrogen (H2) gas. Yet, such responses won’t occur in alkanes and alkenes. The end is that the hydrogen particles connected to the carbon-carbon triple bond in alkynes are somewhat acidic in nature. It is to be noticed the other hydrogen particles exposing these ones are not acidic.
HC ≡ CH + Na → HC ≡ C– Na+ + 1/2H2
Addition Reaction:
Alkynes undergo hydration reactions when the circumstances are right. When alkynes react with halogens, hydrogen, and other similar elements, they produce a saturated molecule. Two hydrogen atoms, halides, or halogens can be added to their structure since they contain a triple bond.
- Addition of Dihydrogen: A catalyst, such as Nickel, Platinum, or Palladium, is used to speed up the process. When hydrogen is added to an alkyne, we get an alkene.
C3H4(g) + 2H2(g) → C3H8(g)
- Addition of Halogens: When alkynes and halogens like chlorine react, the halogen bonds to the alkynes’ structure, resulting in halogen-substituted alkenes. Tetrachloroethane will be the end product.

Addition of halogens
- Addition of Water: Alkynes do not react with water molecules like other hydrocarbons (alkanes and alkenes). This is referred to as immiscibility. A reaction occurs when alkynes are bubbled through dilute sulphuric acid (approximately 40%) in the presence of the catalyst mercuric Sulphate. Carbonyl molecules will be the end products of this process, which is known as a hydration reaction.

Addition of water
- Polymerization: Under the right circumstances, alkynes can polymerize linearly and cyclically. Polymerization occurs, resulting in molecules with a greater molecular weight than the initial alkyne. Ethyne, for example, will polymerize to yield polyacetylene or polyethylene (with a greater molecular weight). High temperatures and the presence of a catalyst are necessary for cyclic polymerization. As in passing ethyne through a red-hot iron tube at a temperature of at least 877K to produce benzene.
Uses of Alkyne
- Since ethyne has an exceptionally hot fire, it is ordinarily utilized in oxyacetylene gas welding and oxyacetylene gas cutting. As ethyne is ignited with oxygen, the subsequent fire is known to have a temperature of around 3600 Kelvin.
- The superseding alkyne in acetylene is utilized as a fuel, with a huge number of kilograms made yearly by partial oxidation of gaseous petrol. Substance mixtures, for example, ethanoic corrosive, acrylic corrosive, and ethanol are produced using a portion of these alkynes.
- Ethyne is most usually used to make natural mixtures like ethanol, ethanoic corrosive, and acrylic corrosive. It’s additionally used to make polymers and crude materials for them.
- Acetylene is separated into two parts, carbon and hydrogen. This response makes a ton of warmth, which can make the gas light regardless of whether there is no air or oxygen present.
- Alkynes are for the most part utilized as the beginning materials for the assembling of an enormous number of natural mixtures of mechanical significance like chloroprene, vinyl chloride and so on.
Sample Questions
Question 1: What is an alkyne?
Answer:
In natural science, alkynes are the unsaturated hydrocarbons having something like a triple connection between carbon-carbon particles (– C≡C–). The overall recipe of this unsaturated hydrocarbon is CnH2n–2, where n–2,3,4,5… … .
Alkynes are customarily known as acetyls. Like different hydrocarbons, alkynes are additionally hydrophobic.
Question 2: What are the properties of alkynes?
Answer:
The first three alkynes are colourless gases, the next eight are liquids, and the higher alkynes are solids. They are odourless, insoluble in water but soluble in organic solvents due to their non-polar nature, have high melting and boiling temperatures, and are lighter than water.
Question 3: What is the main use of alkynes?
Answer:
Acetylene, often known as ethyne, is the most common alkyne chemical. The following are a few examples of applications:
- In gas welding, an oxyacetylene flame is employed.
- Hawker’s lamp and lighthouses both employ acetylene as an illuminant.
- Acetylene is used to make fruits ripen artificially.
- Acetylene is used to make acetaldehydes, acetic acid, ethyl alcohol, synthetic rubber, and other valuable chemicals.
- In organic chemistry, acetylene and its derivatives are commonly employed to make cis and trans alkenes, methyl ketones, and other compounds.
Question 4: How alkynes are acidic in nature?
Answer:
Alkynes have a small negative charge. This is due to the sp hybridization of the triple linked carbon atoms in alkynes, which makes it simpler for them to attract the shared electron pair. Alkyne molecules can readily lose hydrogen as a result. As a result, the acidity of the hydrogen atom connected to the triple bound carbon atom.
Question 5: Why do alkynes have higher boiling points?
Answer:
The melting and boiling temperatures of alkynes rise as the molecular mass of the alkynes increases. Alkynes, on the other hand, have greater melting and boiling points than alkanes and alkenes. This is due to the fact that alkynes are linear and their molecules are packed closer together in space.
Question 6: How are alkynes prepared?
Answer:
Chlorine or bromine is mixed with an inert halogenated solvent like chloromethane to produce a vicinal dihalide from an alkene. To make an alkyne, the produced vicinal dihalide is heated and reacted with a strong base.
Question 7: How can we reduce alkyne to alkene?
Answer:
Alkynes can be transformed to trans-alkenes by dissolving sodium in an ammonia solution. A Na radical gives an electron to one of the P bonds in a carbon-carbon triple bond. This produces an anion in an ammonia solution, which may be protonated by a hydrogen.
Like Article
Suggest improvement
Share your thoughts in the comments
Please Login to comment...